Sarcopenia is common in ulcerative colitis and correlates with disease activity
Article information
Abstract
Background/Aims
Association of sarcopenia with disease severity in ulcerative colitis (UC) is not clearly defined. We planned to estimate the prevalence of sarcopenia in patients with UC as per the revised definition and its relation with the disease severity.
Methods
A cross-sectional assessment of sarcopenia in patients with UC was performed. Disease activity was graded according to complete Mayo score. Hand grip strength was assessed with Jamar hand dynamometer, muscle mass using a dual energy X-ray absorptiometry scan, and physical performance with 4-m walk test. Sarcopenia was defined as a reduction of both muscle mass and strength. Severe sarcopenia was defined as reduced gait speed in presence of sarcopenia.
Results
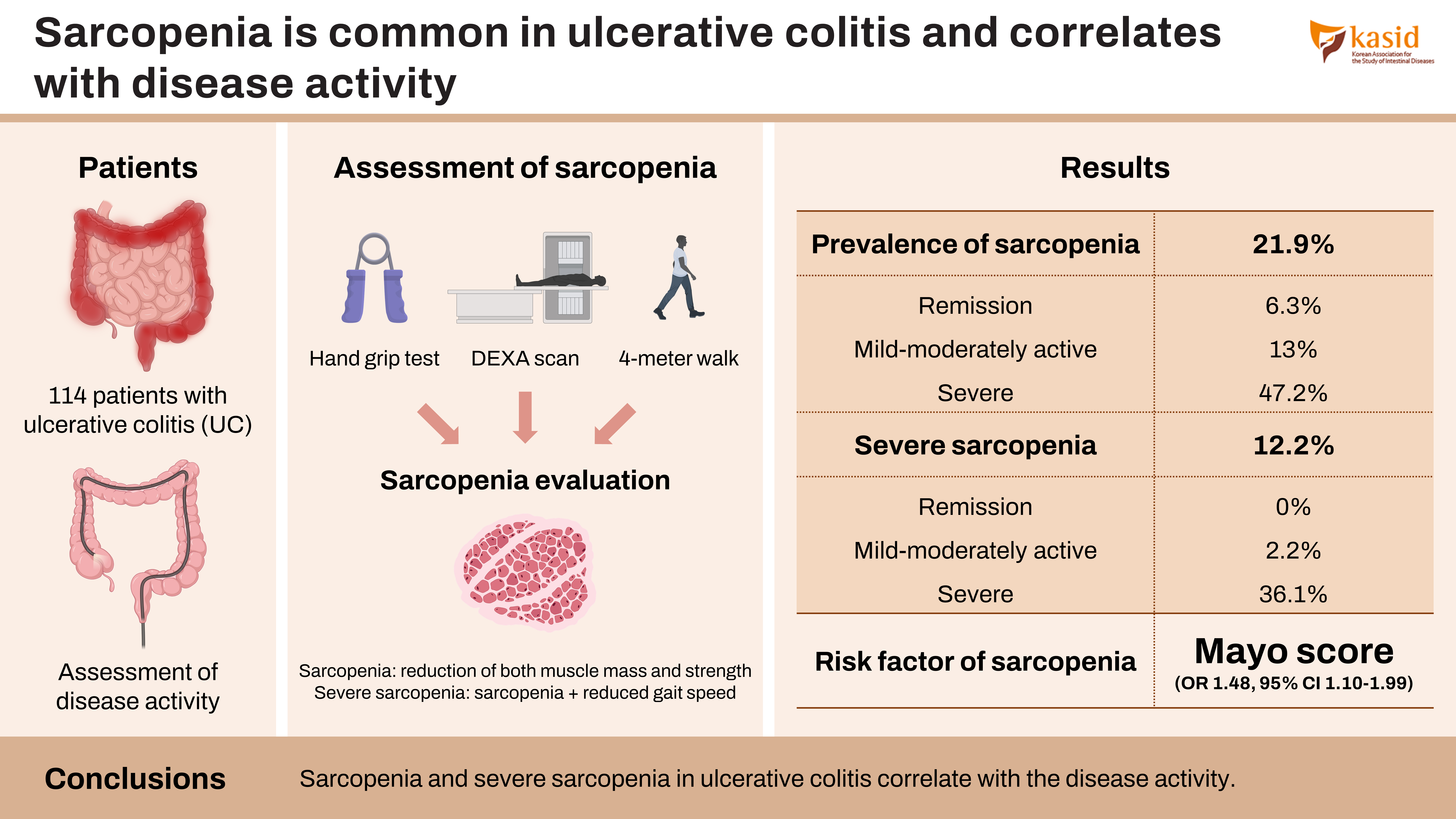
Of 114 patients (62 males, mean age: 36.49±12.41 years), 32 (28%) were in remission, 46 (40.4%) had mild-moderate activity, and 36 (31.6%) had severe UC. Forty-three patients (37.7%) had probable sarcopenia, 25 (21.9%) had sarcopenia, and 14 (12.2%) had severe sarcopenia. Prevalence of sarcopenia was higher in active disease (2 in remission, 6 in active, and 17 in severe, P<0.001). Of 14 with severe sarcopenia, 13 had severe UC while 1 had moderate UC. On multivariate analysis, lower body mass index and higher Mayo score were associated with sarcopenia. Of 37 patients with acute severe colitis, 16 had sarcopenia. Requirement of second-line therapy was similar between patients with and without sarcopenia. On follow-up (median: 18 months), there was a non-significant higher rate of major adverse events in those with sarcopenia (47.4% vs. 33.8%, P=0.273).
Conclusions
Sarcopenia and severe sarcopenia in UC correlate with the disease activity.
INTRODUCTION
Sarcopenia, traditionally a concern in the elderly, is a result of a progressive decline in muscle mass [1]. In recent years, 3 consensus papers were released which provided guidance for defining and diagnosing sarcopenia [2-5]. As per the revised European consensus, probable sarcopenia is identified by the presence of low muscle strength while the diagnosis of sarcopenia is confirmed by the presence of low muscle mass in addition to low muscle strength. Severe sarcopenia is diagnosed if there is low physical performance in presence of sarcopenia.
It is widely recognized now that sarcopenia is not strictly restricted to the elderly age group but is associated with several chronic conditions like chronic liver disease, chronic kidney disease, heart failure, and obstructive airway diseases [6]. Sarcopenia has also been reported with inflammatory bowel disease (IBD). Mechanisms contributing to sarcopenia in IBD include chronic inflammation, malnutrition, malabsorption, and gut dysbiosis through the muscle microbiome axis [7-9]. Data from the systematic review and meta-analyses showed the prevalence of sarcopenia as high as 39.5%. Sarcopenia has also been implicated to have an impact on surgical outcomes in IBD [10-12].
However, most previously reported studies in IBD had various lacunae–most were retrospective, and sarcopenia was defined solely as a reduction in muscle mass. Some recent studies have adapted the consensus definition (using both muscle mass and muscle strength) of sarcopenia and reported the prevalence of sarcopenia in IBD and its impact on surgical outcomes [9,13]. However, the relationship of sarcopenia with disease activity in adult ulcerative colitis (UC) has not been studied. In view of the lacunae in the published literature, we planned this prospective study which was aimed to study the frequency of sarcopenia and severe sarcopenia as per the revised definition in patients with UC. We also planned to report the frequency of sarcopenia in patients with varying disease severity in UC. In addition, we also studied the impact of disease characteristics (including severity, extent, and duration of disease) on sarcopenia and the impact of sarcopenia on the outcomes of hospitalized patients.
METHODS
1. Setting
A prospective observational study was conducted between August 2021 to December 2022 at a tertiary care referral center in Chandigarh, India. Prior approval was taken from the institutional ethics committee, Post Graduate Institute of Medical Education and Research, India vide letter number INT/ IEC/2021/SPL-1578 dated October 25, 2021. All patients provided written informed consent prior to the inclusion. Guidelines on the ethical conduct of human research as provided by the Helsinki Declaration 2013 and the Indian Council of Medical Research 2017 were followed.
2. Patients
All consecutive patients with clinical, endoscopic, and radiological features suggestive of IBD presenting either to the outpatient department or admitted to the emergency, wards of the institute were screened for possible inclusion into the study. All patients with diagnosis of UC were considered for inclusion in the study. The diagnosis of UC was based on a combination of clinical, endoscopic, and histologic criteria as outlined in the European Crohn’s and Colitis Organisation guidelines [14]. We excluded patients who were ≤ 18 years, pregnant or lactating women, those with additional risk factors for sarcopenia (chronic kidney disease, congestive heart failure, cerebrovascular accidents, chronic obstructive pulmonary diseases, chronic liver disease or cirrhosis, diabetes mellitus, muscular dystrophy, and amputations), and those refusing consent. Demographic details, clinical history, past and current drugs, hospitalization status and socio-economic status based on a modified Kuppuswamy scale were recorded [15]. Each patient underwent a physical examination including an anthropometric assessment. Blood investigations including tests which would influence muscle health were collected including complete blood counts, vitamin D levels, serum calcium, total cholesterol, and serum albumin levels.
3. Assessment of Disease Activity
Disease activity was measured with the Mayo score for UC. Following parameters for each patient were recorded, at the point of recruitment in the study, which includes stool frequency, amount, and frequency of blood in stools, endoscopic disease activity, and a physician’s global assessment. Each variable was given a score from 0 to 3 and the final score was taken as the sum of individual scores. Extent of disease was classified according to Montreal classification into E1 for proctitis, E2 for left-sided colitis involving till splenic flexure and E3 for disease beyond splenic flexure [16]. The patients were followed up for the occurrence of any major adverse events (MAE; flare of disease, requirement of steroids or advanced therapies, hospitalization, surgery) in the follow-up period.
4. Assessment of Sarcopenia and Its Severity
1) Hand Grip Strength Testing
Hand grip strength (HGS) was assessed using a handheld dynamometer (Jamar hydraulic hand dynamometer). Each patient was asked to sit comfortably in a chair with shoulder adducted, elbow in 90° flexion, and wrist at 0° to 30° dorsiflexion and 0° to 15° ulnar deviation position. The handlebars were adjusted according to the patient’s comfort level. Each patient was asked to press the handle as tightly as possible for a minimum of 5 seconds, a total of 3 recordings were taken from each arm alternatingly. Taking fatigue into consideration a gap of a minimum of one minute is given between each recording. A mean of 3 recordings was taken. The patient’s dominant hand grip values were compared to cutoff values derived from healthy cohorts of the local population and hand grip is considered reduced if it was < 27.5 kg in males and < 18 kg in females [17].
2) Assessment of Muscle Mass
Muscle mass was assessed using dual energy X-ray absorptiometry (DEXA) scan, we have used Hologic Discovery A DEXA (Hologic, Inc., Bedford, MA, USA). All scans were done by a dedicated, certified technician. Each patient underwent an anthropometry assessment including height and weight before the DEXA scan following which a whole-body scan was done, muscle mass was assessed by the appendicular skeletal mass derived from the DEXA scan following which appendicular skeletal muscle index (ASMI) was calculated by dividing appendicular skeletal mass with height in meters square. It was done to standardize muscle mass with height as short persons would have lesser overall muscle mass when compared to taller persons. The derived ASMI values were compared with the healthy cohort of the local population and ASMI was considered reduced if it was < 6.11 kg/m2 in males and < 4.61 kg/m2 in females [17].
3) Assessment of Muscle Function
Physical performance was assessed with a 4-m walk test, a 4-m distance was marked with a meter tape and the patient was asked to walk the distance between the 2 points at his comfortable speed, time was recorded with a stopwatch and 2 readings were taken. Gait speed (m/sec) was calculated by dividing distance (4 meters) by time (seconds) and the mean of the 2 recordings was taken as the gait speed for the study. Gait speed is considered reduced if it was < 0.8 m/sec [17,18].
4) Definitions of Sarcopenia and Severe Sarcopenia
Probable sarcopenia was defined as the presence of reduced grip strength. Sarcopenia was defined as the presence of both reduced grip strength and muscle mass. Severe sarcopenia was defined as the presence of sarcopenia with reduced gait speed [18]. The patient was defined to be in remission if the Mayo score was ≤ 2. The patient was considered to have severe disease if the Mayo score was ≥ 11 or required admission. Patients with a Mayo score of 3–10 and not requiring hospitalization for disease activity were considered to have mild to moderately active disease [19].
5. Statistical Analysis
Data analysis was done in SPSS software version 22.0 (IBM Inc., Armonk, NY, USA). The normality of data was assessed using the Kolmogorov-Smirnov test. Qualitative variables were expressed as proportions. Quantitative variables were expressed as either mean or median, with standard deviation and interquartile range as measures of dispersion respectively. Each patient was stratified according to the disease severity and the prevalence of probable sarcopenia, sarcopenia and severe sarcopenia was calculated and compared among the groups with the Pearson chi-square test or Mann-Whitney U-test and a P-value of < 0.05 was considered significant. A receiver operating characteristic (ROC) curve was calculated for different points across the Mayo score to accurately predict the presence of sarcopenia or severe sarcopenia and the reference point was taken at which both the sensitivity and specificity were maximum. A multivariate logistic regression analysis was carried out for sarcopenia to find out the variables associated with the presence of sarcopenia. For admitted patients with severe UC, as defined by Truelove-Witts’s criteria, the prevalence of probable sarcopenia, sarcopenia and severe sarcopenia was compared among patients who improved with standard therapy with patients who required second-line therapy, using the Fischer exact test and the P-value of < 0.05 was considered significant [20].
RESULTS
1. Patients
A total of 168 patients were assessed for eligibility of which 114 patients with UC were included in the final analysis (Fig. 1). The mean age of the study group was 36.49 ± 12.41 years. Sixty-two patients (54.4%) were males. Most of them belonged to rural localities (n = 78, 68.4%) and were from lower-middle and upper-lower socio-economic strata according to the modified Kuppuswamy scale (n = 35 [30.7%] and n = 33 [28.9%], respectively). Anthropometric measures including mean triceps skin fold thickness and body mass index (BMI) levels were 6.60 ± 3.27 mm and 20.90 ± 4.02 kg/m2, respectively. Biochemical measures including mean hemoglobin, neutrophil-lymphocyte ratio (NLR), albumin, vitamin D, calcium, and total cholesterol levels were 10.27 ± 2.56 g/dL, 3.90 ± 2.72, 3.66 ± 0.98 g/dL, 19.60 ± 11.17 ng/mL, 8.90 ± 0.56 mg/dL, and 135.27 ± 51.60 mg/dL respectively.
2. Disease Variables and Severity
The mean duration of disease was 45.7 ± 50.7 months. Most of the patients (n = 58, 50.9%) had a duration between 1 to 5 years, followed by < 1 year duration (n = 30, 26.3%) and > 5-year duration (n = 26, 22.8%) respectively. Disease extent was studied in 98 patients, E3 was the most prevalent disease found in 55 patients (56.8%) followed by E2 in 34 patients (34.7%) and E1 was found in 9 patients (9.1%). Sixty-one patients (53.5%) had past hospital admission, 44 patients (38.6%) were admitted during the evaluation period and 9 patients were never hospitalized. Of 44 admitted during the study period, 37 had acute severe UC as per modified Truelove and Witts criteria. At the time of inclusion, 32 (28%) patients were in remission, 46 patients (40.4%) had active disease and 36 patients (31.6%) had severe disease. Mean C-reactive protein (CRP) levels in the study group were 20.1 ± 26.6 mg/L. During the study period, 106 (93.8%) and 64 (56.6%) were on oral and rectal 5-aminosalicyclic acid preparations respectively. Fifty-two patients (46.0%) were on thiopurine (azathioprine). Forty-nine patients (43.4%) were on steroids at the time of inclusion into the study for a short duration to take care of the active UC and steroid use is merely a marker of active UC requiring steroids. In all these patients, the duration of use was < 14 weeks. Three patients were on tofacitinib, 9 were on biologics, and 6 received cyclosporine during hospitalization. All the baseline variables have been summarized in Table 1.
3. Sarcopenia Variables
Overall mean dominant HGS was 27.1 ± 10.2 kg, it was 20.6 ± 6.2 kg in females and 32.5 ± 9.8 kg in males. Among them, 19 females (36.5%) and 24 males (38.7%) had reduced dominant HGS (P= 0.812). The mean ASMI in the study group was 5.64 ± 1.54 kg/m2 (females 5.31 ± 1.83 kg/m2 and males 5.92 ± 1.20 kg/m2). A total of 15 females (28.8%) and 37 males (59.7%) were considered to have low muscle mass (P= 0.001) based on ASMI. The mean gait speed in the 4-m walk test was 0.95 ± 0.14 m/s, 35 patients (30.7%) had a gait speed of < 0.8 m/s.
1) Prevalence of Sarcopenia and Severe Sarcopenia
Forty-three patients (37.7%) had probable sarcopenia among them, 25 (21.9%) and 14 (12.2%) were found to have sarcopenia and severe sarcopenia, respectively.
2) Sarcopenia with Respect to Disease Activity
There was a moderately positive correlation between the Mayo disease activity and the presence of probable sarcopenia, sarcopenia, and severe sarcopenia with r values of 0.481, 0.338, and 0.433 respectively, consecutively there was a negative correlation between the Mayo disease activity with variables of sarcopenia, i.e., HGS, ASMI, and gait speed (r= –0.451, r= –0.207, r= –0.426, respectively) (Fig. 2). When stratified according to Mayo scores, probable sarcopenia was present in 5 (15.6%), 11 (23.9%), and 27 (75%) patients who were in remission, mild to moderately active and severe disease respectively and the difference was statistically significant (P< 0.001). Sarcopenia was present in 2 (6.3%), 6 (13%), and 17 (47.2%) patients who were in remission, mild-moderately active and severe disease respectively and the difference was statistically significant (P< 0.001) (Table 2). A Mayo score of > 8 was 84% sensitive and 63% specific in predicting the presence of sarcopenia (area under the ROC curve = 0.773) (Supplementary Fig. 1A). None of the patients in remission had severe sarcopenia, one patient with mild to moderately active disease and 13 patients (36.1%) with severe disease had the presence of severe sarcopenia with a significant difference among the groups (P< 0.001) (Table 2). A Mayo score of > 8 was 100% sensitive and had a specificity of 60% in predicting the presence of severe sarcopenia (area under the ROC curve = 0.870) (Supplementary Fig. 1B).

Correlation of HGS (A), ASMI (B), and gait speed (C) with disease activity (complete Mayo score). HGS, hand grip strength; ASMI, appendicular skeletal muscle index.
3) Relationship between Disease Variables and Sarcopenia
We found out that sarcopenia was more prevalent among patients with a disease duration of ≤ 1 year (n = 14, 46.6%) compared to those with a duration of > 1 year (n = 11, 13.1%) to explain this finding we have stratified the disease activity as per the Mayo score and found out that more patients with disease duration of > 1 year were in remission (n = 27, 32.1%) or had mild to moderately active disease (n = 38, 45.2%). There were a greater number of patients with severe disease (n = 17, 56.6%) in those with disease duration ≤ 1 year. Suggesting that sarcopenia was associated with disease severity rather than disease duration (Supplementary Table 1).
4. Sarcopenia and Outcomes in Hospitalized Patients
Out of 114 patients, 37 patients were admitted with acute severe colitis as per Truelove and Witts criteria requiring IV steroid therapy. Twenty-six patients (70.2%) responded to steroids and the rest 11 patients (29.8%) received second-line therapy, in which 6 received cyclosporine, 1 received biologics (infliximab), 3 received tofacitinib, 1 underwent colectomy. The prevalence of sarcopenia and severe sarcopenia among admitted patients were 16 (43.2%) and 12 (32.4%), respectively. Among those with sarcopenia, 3 patients (18.7%) required second-line therapy as compared to 8 (38%) in those without sarcopenia (P= 0.202). Out of 12 patients with severe sarcopenia, 2 (16.7%) received second-line therapy compared to 8 (32%) patients without severe sarcopenia (P= 0.228) (Supplementary Table 2).
1) Follow-up Data on MAE
Follow-up data was available for 93 patients (males = 50, 53.8%) with a median follow-up of 18 months (interquartile range, 15–20 months). The occurrence of any MAE (a composite measure of hospitalization or need for steroids/advanced therapy/ colectomy) was noted in 34 patients (36.5%). A higher trend of MAE was seen among those with sarcopenia as compared to those without sarcopenia (47.4% vs. 33.8%, P= 0.273) (Supplementary Table 3).
2) Multivariate for the Prediction of Sarcopenia
On univariate analysis, the baseline parameters found to be statistically significant in predicting sarcopenia were disease duration (P=0.039), smoking (P=0.026), steroid usage (P=0.004), triceps skin fold thickness (P= 0.045), BMI (P< 0.001), hemoglobin (P= 0.003), NLR (P= 0.030), albumin (P= 0.002), CRP (P= 0.018), and Mayo score (P< 0.001). On multivariate analysis, only BMI (odds ratio, 0.622; 95% confidence interval, 0.457–0.847; P= 0.003) and Mayo score (odds ratio, 1.485; 95% confidence interval, 1.107–1.991; P= 0.008) had a statistically significant association with sarcopenia (Table 3).
DISCUSSION
In this prospective cross-sectional study on consecutive cases of UC, the prevalence of sarcopenia was 21.9%. Sarcopenia was found to be higher in patients with active disease. Patients with sarcopenia had longer disease duration. Mayo score could predict the presence of sarcopenia and severe sarcopenia. Patient variables including the history of smoking and steroid usage, presence of low BMI, triceps skinfold thickness, hemoglobin, albumin; high NLR, and CRP were associated with sarcopenia. However, only BMI and Mayo score were found to be predictors of sarcopenia in multivariate analysis. The study, to the best of our literature search, the first study to use the revised definition of sarcopenia to define the frequency in a South Asian cohort and to provide evidence of relationship between sarcopenia and disease activity.
The prevalence of sarcopenia in our study was 21.9%, which was high when compared to the cross-sectional study of 222 and 42 UC patients where the prevalence was 5.8% and 14% respectively [21,22]. The probable reason might be due to preexisting malnutrition as most of the patients in our study group belonged to the lower socio-economic class. Severe sarcopenia was seen in 12.2% of patients in our study. To date, no studies have reported the individual prevalence of severe sarcopenia in UC patients. Till 2019 most of the studies have explored the association of myopenia with the outcomes and surgical complications in IBD patients [10,23-25]. There were only a few prospective studies which have adopted the consensus definition of sarcopenia, but these studies did not correlate disease activity with the prevalence of sarcopenia [9,21,22].
We have noticed that the prevalence of sarcopenia in hospitalized UC patients was high (43.2%) when compared to the overall prevalence (21.9%) which can be explained by the disease activity as the mean Mayo score in hospitalized patients was higher (11.03 ± 0.45 vs. 6.57 ± 4.00) a similar prevalence (48.27%) was noted by Bamba et al. [26], in 29 hospitalized patients with UC. Cushing et al. [27], in their study on 89 patients with acute severe UC (ASUC) demonstrated the requirement of rescue therapy was higher in patients with sarcopenia (56% vs. 28%, odds ratio = 3.98). However, sarcopenia was considered as a reduction in muscle mass only and ASUC was not defined as per Truelove and Witts criteria which were addressed in our study. In contrast to their finding, there was no difference in the response to intravenous corticosteroid (IVCS) therapy among the 37 cases of ASUC, this can be explained by the high response rates to steroids in UC among the South Asian cohort and because we classified sarcopenia as per the consensus definition [28]. Another study by Ge et al. [25], in 233 patients of ASUC defined as per Truelove and Witts showed that the presence of sarcopenia (defined as a reduction in muscle mass) was a predictor of failure to IVCS. However, the overall response rate to IVCS in this study cohort was only 35.2% which was much lower than the expected response (67%) and hence could not be compared to our cohort [14]. The higher steroid response is expected in our population which largely naïve to biologicals and small molecules.
Sarcopenia is often overlooked and undertreated part, growing evidence shows that sarcopenia is associated with increased mortality in diseases like cirrhosis, and chronic kidney disease to name a few [29,30]. Our results have shown that steroid usage was associated with sarcopenia but it cannot be explained if it was related to the drug or underlying disease activity, as patients with active disease required steroids, the cause and effect of this relationship have to be explored in future cohort studies. Observational data suggest that therapy with biologics (infliximab) in Crohn’s disease had a reversal of sarcopenia and further evidence in UC may help us choose appropriate targeted therapy [31]. Our study identified that those with active disease may be at heightened risk for sarcopenia and should be followed up with a measure of muscle-related parameters once the disease activity is controlled. Extrapolating the result of our study, screening for sarcopenia can be considered in patients with active disease. Follow-up data of our cohort showed that the prevalence of MAE had a slightly higher trend in those with sarcopenia hinting that interventions directed at sarcopenia might have a benefit on disease activity. It remains to be seen if interventions aimed at treating sarcopenia could improve sarcopenia and clinical outcomes in these patients.
There are a few limitations to our study first it was a crosssectional observational study and patients could not be followed up to assess the readmission rates and response rates, especially in the setting of outpatient care. Changes in sarcopenia with respect to therapy and reduction in disease activity could not be followed up prospectively and the impact of sarcopenia on the quality of life could have been measured. Functional status (4-m walk) cutoff was defined for the elderly cohort which we have used in our study is a limitation and we acknowledge it. However, there are strengths to our study, we have evaluated and diagnosed sarcopenia based on the consensus definition. We have compared the prevalence of sarcopenia with disease activity and showed that disease activity was an important risk factor for sarcopenia, and we have followed up patients for any new MAE to assess the impact of sarcopenia at the baseline on the disease-related outcomes. We have addressed the issue of sarcopenia in IBD in a South Asian cohort-a region widely recognized as a new frontier of IBD [32].
This study was set out to find out the prevalence and severity of sarcopenia among patients with UC and its relation with disease activity. Prevalence of sarcopenia was higher in patients with active disease and is influenced by disease activity with a significantly higher proportion of patients with mild to moderately active and severe disease having probable sarcopenia, sarcopenia, and severe sarcopenia. However, the presence of sarcopenia did not seem to influence the outcome of therapy and the requirement of second-line therapy in those with ASUC. These findings illustrate that sarcopenia is common in patients with active disease and is often an ignored entity. Future longitudinal studies with larger sample sizes are required to study the impact of sarcopenia on the outcomes, impact on the quality of life and outcomes in intervening sarcopenia with respect to disease activity and quality of life.
Notes
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Data Availability Statement
The anonymized data can be obtained from the corresponding author on a reasonable request.
Author Contributions
Conceptualization: Sharma V. Data curation; Formal analysis: Sharma V, Neelam PB. Investigation; Methodology: Neelam PB, Pal R, Sekar A, Bhadada SK, Gupta P, Sharma V. Project administration: Sharma V, Dutta U. Resources: Singh AK, Gupta P, Shah J, Mandavdhare HS, Sharma V, Singh H, Dutta U. Software: Neelam PB. Supervision; Validation: Sharma V, Bhadada SK, Dutta U. Visualization: Neelam PB, Sharma V. Writing - original draft: Neelam PB. Writing - review & editing: Sharma V. Approval of final manuscript: all authors.
Supplementary Material
Supplementary materials are available at the Intestinal Research website (https://www.irjournal.org).
Supplementary Table 1.
Sarcopenia with Respect to Disease Duration
Supplementary Table 2.
Outcomes with Respect to Sarcopenia in Hospitalized Patients with Acute Severe Ulcerative Colitis
Supplementary Table 3.
Major Adverse Events in Patients on Follow-up
Supplementary Fig. 1.
(A) Receiver operating characteristic (ROC) analysis of prediction of sarcopenia using Mayo score. (B) ROC analysis of prediction of severe sarcopenia using Mayo score.