Distribution of cytomegalovirus genotypes among ulcerative colitis patients in Okinawa, Japan
Article information
Abstract
Background/Aims
To determine the prevalence of glycoprotein B (gB), glycoprotein N (gN), and glycoprotein H (gH) genotypes of human cytomegalovirus (HCMV) superimposed on ulcerative colitis (UC) patients in Japan.
Methods
Four archived stool samples and 7-archived extracted DNA from stool samples of 11 UC patients with positive multiplex polymerase chain reaction (PCR) results for HCMV were used UL55 gene encoding gB, UL73 gene encoding gN, and UL75 gene encoding gH were identified by PCR. Genotypes of gB and glycoprotein N were determined by sequencing.
Results
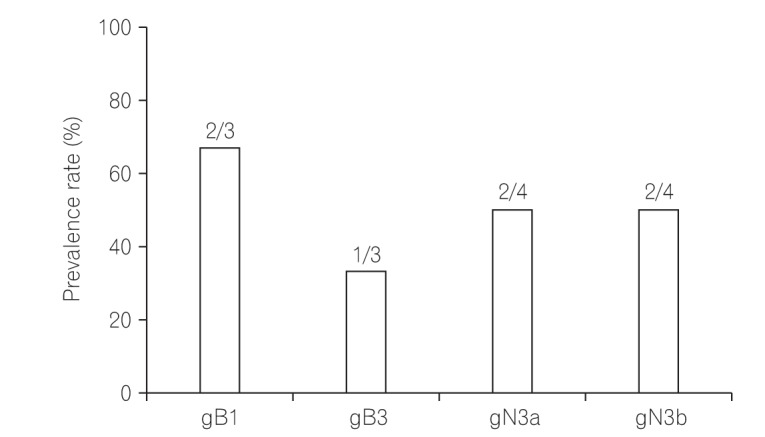
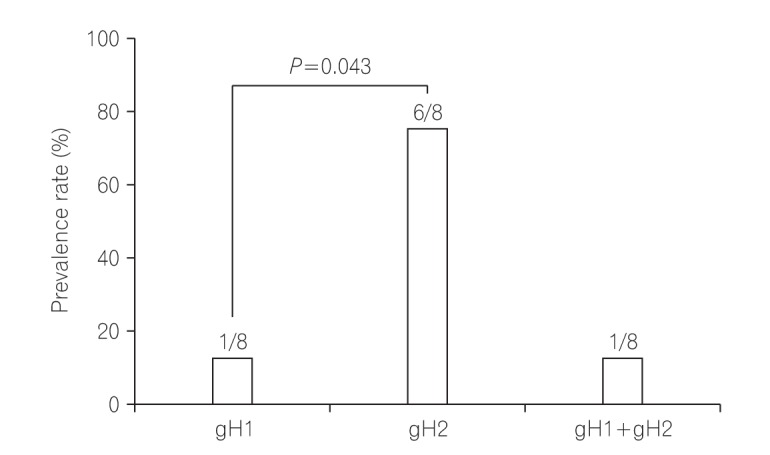
Among 11 samples, 8 samples were amplified through PCR. gB, gN, and gH genotypes were successfully detected in 3 of 8 (37.5%), 4 of 8 (50%), and 8 of 8 (100%), respectively. The distribution of gB and gN genotypes analyzed through phylogenetic analysis were as follows: gB1 (2/3, 66.7%), gB3 (1/3, 33.3%), gN3a (2/4, 50%), and gN3b (2/4, 50%). Other gB genotypes (gB2 and gB4) and gN genotypes (gN1, gN2, and gN4) were not detected in this study. Out of successfully amplified 8 samples of gH genotype, gH1 and gH2 were distributed in 12.5% and 75% samples, respectively. Only 1 sample revealed mixed infection of gH genotype. The distribution of gH1 and gH2 differed significantly (1:6, P<0.05) in UC patients. The distribution of single gH genotype also revealed significant difference in UC patients who were treated with immunosuppressive drug (P<0.05).
Conclusions
In this study, gB1, gN3, and gH2 gene were determined as the most frequently observed genotypes in UC patients, which suggest that there might be an association between these genotypes of HCMV and UC.
INTRODUCTION
The human cytomegalovirus (HCMV) genome is the largest of all human herpes viruses and is a linear 220- to 240-kbp double-stranded DNA comprising more than 200 open reading frames.1 Certain genes within the HCMV genome exhibit genetic variability, thus enabling the differentiation of genetically distinct HCMV genotypes. Genotyping targets of HCMV mainly focus on envelope glycoproteins. HCMV genes encoding glycoproteins often show genetic polymorphism. Several envelope glycoproteins such as UL55 gene encoding glycoprotein B (gB), UL73 gene encoding glycoprotein N (gN), UL75 gene encoding glycoprotein H (gH), UL74 gene encoding glycoprotein O (gO), and UL115 gene encoding glycoprotein L (gL) have been evaluated in clinical isolates due to their significant role in tissue tropism and virulence.23456 It has been hypothesized that genetic variation among HCMV strains may underlie strain-specific clinical manifestations. Four gB genotypes have been identified based on sequence variation in the UL55 gene encoding gB: gB1, gB2, gB3, and gB4.7 gN is also highly polymorphic at the amino-terminal region. Most clinical HCMV isolates cluster into 4 gN genomic variants (gN1, gN2, gN3, and gN4), and gN3 and gN4 are further subdivided into the gN3a and gN3b and gN4a, gN4b, gN4c, and gN4d genotypes, respectively.8 With respect to gH genotypes, gH1 and gH2 have been identified among clinical HCMV isolates.9 gB, gN, and gH have been studied extensively in both immunocompetent and immunocompromised patients. Genotyping of HCMV has revealed significant differences in the pathogenicity of specific strains in congenitally infected newborns and immunocompromised patients.101112
UC patients are often considered immunocompromised due to complications resulting from disease progression or the use of immunosuppressive drug therapy (such as corticosteroids, immunomodulatory drugs, and anti-tumor necrosis factor). Indeed, UC patients undergoing intensive immunosuppression frequently experience reactivation of HCMV. Consequently, colonic reinfection or reactivation of latent HCMV is considered important exacerbating factors in steroid-dependent and steroid-refractory UC patients.13 However, some researchers claim HCMV does not contribute to increased morbidity and mortality in UC.14 Differences among gB, gN, and gH strains may explain the differences in UC patient outcomes. Therefore, we aimed to determine the frequency distribution of the gB, gN, and gH genotypes and to explore the relationship between these genotypes and the clinical characteristics and outcome of UC patients.
METHODS
1. Study Population and Specimens
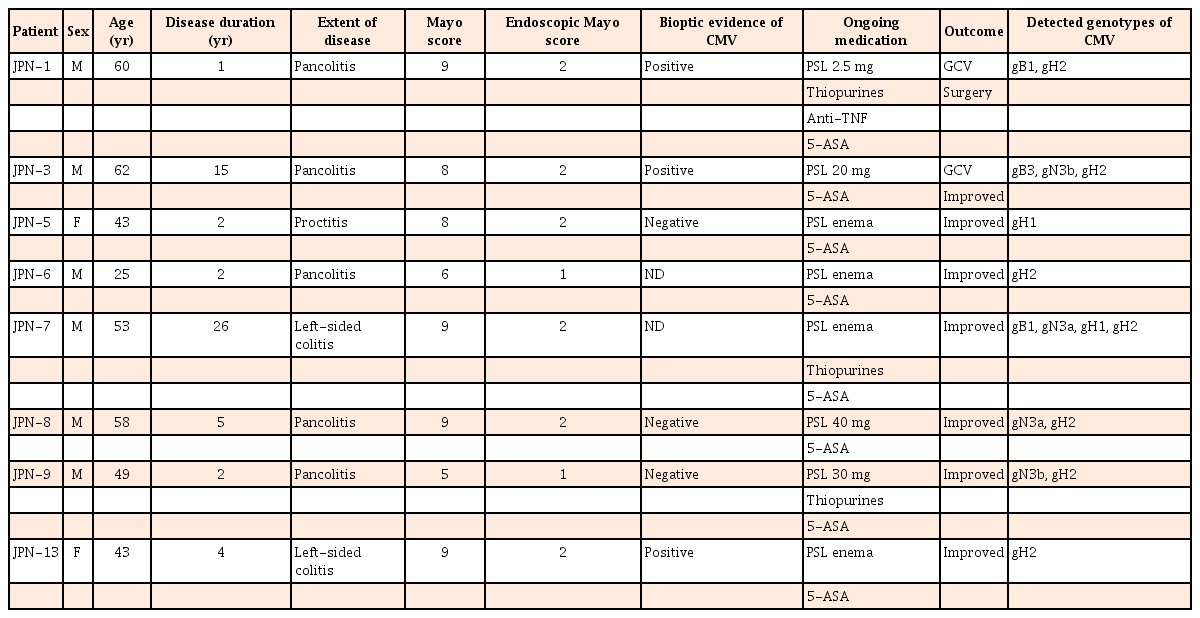
In our prior study,15 we prospectively analyzed 300 patients who underwent colonoscopy at the Department of Endoscopy at the University of the Ryukyus Hospital from August 2014 to January 2015. Stool samples were collected endoscopically from the recto-sigmoid region of the colon and were transported to the laboratory. Stool samples were immediately processed for the multiplex PCR. Among above patients and specimens, 11 adult UC active patients with HCMV infection were retrospectively analyzed from archived samples (n=4) or archived extracted DNA (n=7) stored at −80℃. HCMV-positive results were determined previously using the Seeplex Meningitis V1 multiplex PCR kit (Seegene Inc., Seoul, Korea). HCMV virus diversity was evaluated by genotyping the envelope glycoproteins UL55 (gB), UL73 (gN), and UL75 (gH) in HCMV-positive UC patients. Additional clinical and laboratory data were collected from medical records. The characteristics of the patients were evaluated with factors including, disease duration, extent of disease, Mayo score (scores can range from 0 to 12, with higher scores indicating more severe disease activity),16 Mayo endoscopic subscore, bioptic evidence of cytomegalovirus (CMV), medication, and outcome. This prospective study was approved by the Institutional Review Board of the University of the Ryukyus, and written informed consents were obtained from all patients prior to their inclusion in the study.
2. Sample Preparation for PCR
DNA extraction was performed using the Ribospin™ vRD kit (GeneAll Biotechnology, Seoul, Korea) according to the manufacturer's instructions. All extracted DNA utilized in this study was stored at −80℃.
3. Characterization of the CMV gB, gH, and gN Genotypes
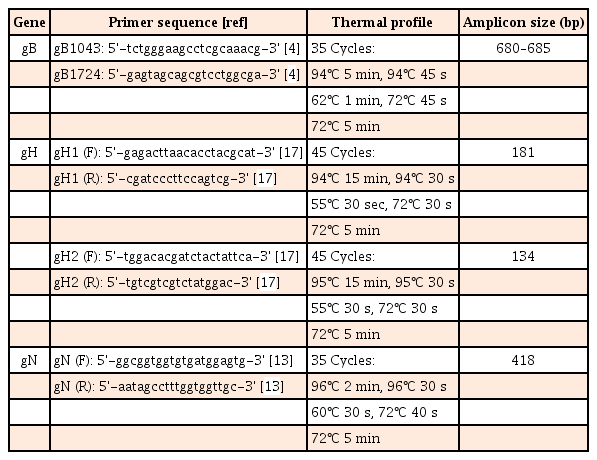
After confirming HCMV by multiplex PCR, the 11 DNA extracts were subjected to single-target PCR to amplify UL55 (gB), UL73 (gN), and UL75 (gH1 and gH2) as gene targets for sequencing. The primers and thermal conditions for amplification are listed in Table 1.41317 Purification and sequencing of the successfully amplified products of gB and gN were performed by Hokkaido System Science Co., Ltd. (Hokkaido, Japan) and sent the chromatograms.
4. Phylogenetic Analysis
Phylogenetic analysis to further characterize the gB and gN genotypes was performed using strain sequences retrieved from GenBank. The GenBank accession numbers of the UL55 (gB) and UL73 (gN) gene sequences used for comparison were as follows: M60929.1 for gB1, M60931 for gB2, M85228.1 for gB3, M60926.1 for gB4; AF309969 for gN1, AF309975 for gN2, AF309980 for gN3a, AF390835 for gN3b, AF309988 and AF390848 for gN4a, AF390797 and AF390857 for gN4b, AF390748 for gN4c, and AF310006 for gN4d. Sequences were aligned using CLUSTALW (http://www.genome.jp/), and the resulting phylogenetic tree was generated by the maximum likelihood method using MEGA 6.0 (http://www.megasoftware.net).
5. Statistical Analysis
Statistical analysis was performed using SPSS software version 18.0 (SPSS Inc., Chicago, IL, USA). Categorical variables were compared using the chi-square test in a 2×2 contingency table. P-values of less than 0.05 were considered statistically significant.
RESULTS
Eleven stool specimens from UC patients with positive multiplex PCR results for HCMV were analyzed to determine the genotypes of the 3 major genes, that is, the UL55 (gB), UL73 (gN) and UL75 (gH) genes. These genotypes were successfully amplified in 8 of 12 cases and PCR amplification was unsuccessful in 4 of 12 cases. All successfully amplified products contained gH1 and gH2 (8, 100%), 4 contained gN (50%), and 3 contained gB (37.5%) (Fig. 1).
Gel images of PCR product of (A) gH, (B) gB, and (C) gN genotypes of human cytomegalovirus respectively after analyzing through electronic electrophoresis system. gH, glycoprotein H; gB, glycoprotein B; gN, glycoprotein N; LM, lower marker; UM, upper marker; JPN, Japan.
1. Patients Characteristics
Clinical and endoscopic characterization of UC patients who were participated in this study is shown in Table 2. Successfully genotyped 62.5% of patients (5/8) had pancolitis, 25% (2/8) had left-sided colitis, and 12.5% (1/8) had proctitis. Male to female ratio was 3:1 (male, 6; female, 2) and the average age was 49.13 years. Eighty-seven point five percent of UC patients (7/8) experienced severe colitis and 12.5% of patients had moderate activity revealed by Mayo score.
2. With Regard to gB Genotypes of CMV
The samples containing gB most frequently clustered nearer to other gB1 sequences (2/3, 66.7%), followed by gB3 (1/3, 33.3%). Genotypes gB2 and gB4 were not observed in UC active patients (Figs 2 and 3).
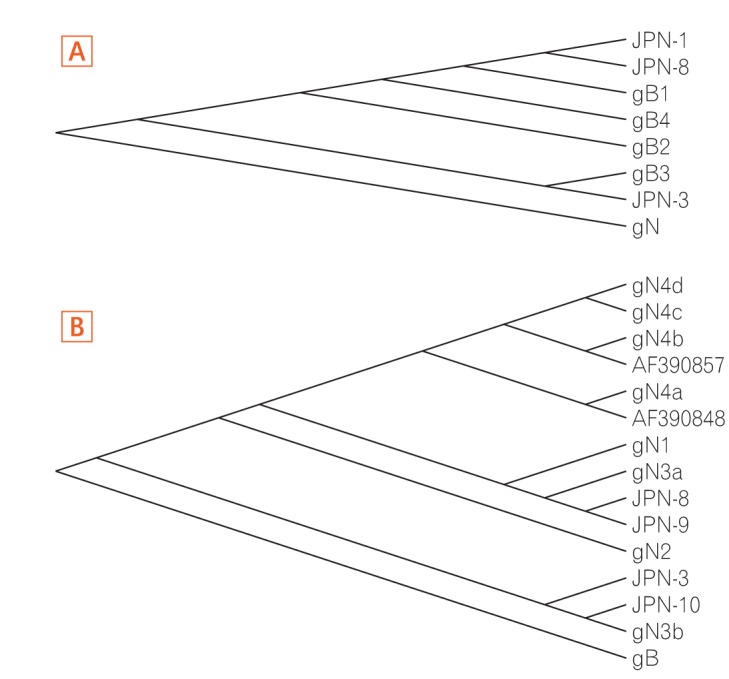
Phylogenetic analysis of glycoprotein B (gB) and glycoprotein N (gN) genotypes of cytomegalovirus. The reference strains and isolates detected through NCBI BLAST are inserted for genetic comparison. The phylogenetic tree (rectangular) with 1,000 bootstrap replicates was reconstructed using maximum likelihood method and Kimura 2-parameter distances. (A) and (B) represent genetic relationships of gB and gN genotypes of cytomegalovirus, respectively. The strains detected in this study are represented by sample identification with codes mentioned as JPN: Japan.
3. With Regard to gN Genotypes of CMV
All gN samples (4/4) showed the gN3 genotype in phylogenetic analysis, with 2 clustering within gN3a and 2 clustering within gN3b. The other gN genotypes (gN1, gN2, gN3c, and gN4) were not detected in this cohort (Figs 2 and 3).
4. With Regard to gH Genotypes of CMV
The prevalence and distribution of HCMV glycoproteins are shown in Fig. 4. The HCMV UL75 amplicons were detected in all 8 successfully amplified stool samples. The gH1 and gH2 genotypes were distributed in 12.5% (1/8) and 75% (6/8) of the samples, respectively. The gH2 genotype was the most frequently observed. The distributions of the gH1 and gH2 genotypes differed significantly (1:6, P=0.043). Only 1 of 8 patients (12.5%) was observed to have a mixed gH genotype infection. In immunosuppressed patients alone, gH2 genotype was observed more frequently.
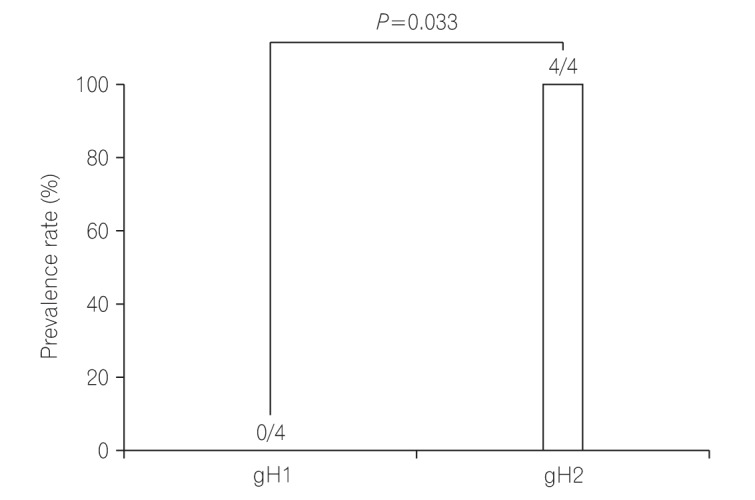
Among gH gene-positive UC patients, 5 patients were treated with immunosuppressive drugs (steroids, thiopurines, anti-tumor necrosis factor). Among these 5 patients, a single gH gene was observed in 4 of 5 (80%) and mixed gH genotypes were observed in 1 of 5 (20%). gH2 was observed in all 4 single gH gene-positive UC active patients receiving immunosuppressive drugs, with no detection of gH1. In statistical analysis, a significant difference was observed in the distribution of single gH genotypes among UC patients receiving immunosuppressive drugs (P=0.033) (Fig. 5). The relationship between clinical features and gH genotype was investigated in this study. gH2 genotype showing 62.5% of patients (5/8) had pancolitis and 12.5% of patients (1/8) had left sided colitis (Table 2).
DISCUSSION
HCMV infection or reactivation in healthy individuals is typically asymptomatic. However, in immunocompromised patients, symptoms may develop in multiple organs and include a variety of diseases, such as retinitis, pneumonia, and intestinal disease, primarily colitis.181920 Emerging evidence also suggests that HCMV may play a significant role in the exacerbation of UC and cause significant morbidity and mortality in patients with induced immunodeficiency. Many studies have reported that immunosuppressive drugs in IBD patients might play a role in the reactivation of HCMV, which in turn may further exacerbate IBD symptoms, particularly in UC patients.1321 Using multiplex PCR, we previously detected significantly more HCMV-DNA in stool samples of UC patients compared with non-UC patients. We further indicated that immunosuppressive drugs and the disease characteristics of UC itself may increase the risk of HCMV reactivation in UC patients.15 However, another theory was that HCMV does not play a pathogenic role in UC patients.22 The observed pathogenic diversity might be due to pathogenic and non-pathogenic genotypes of HCMV.23 Therefore, it is important to determine the differences between non-pathogenic and pathogenic strains.
Genetic polymorphisms among HCMV strains may correlate with important clinical characteristics involved in HCMV disease etiology. Indeed, several studies have shown that genetic variability among HCMV strains may play an important role in immunopathogenesis and disease progression.24
Very little is known about the distribution of HCMV glycoprotein genes in UC patients. In the present study, we focused on gB, gN, and gH genotypes of cytomegalovirus to illuminate the prevalence rate in this cohort. Based on the significant genetic variation observed in these genes, the gB, gN, and gH genotypes of HCMV are good candidates to illustrate differences in pathogenicity.
This is the first study to focus on the gB, gN, and gH genes of HCMV simultaneously in UC patients. Previous studies have evaluated the prevalence rates of several genotypes of HCMV genes in patients with congenital infection or other populations of immunocompromised patients, such as human immunodeficiency virus-positive patients, bone marrow transplant recipients, renal transplant patients, and solid organ transplant patients.25262728 With respect to the gB genotype, previous studies have reported that gB1 is the most frequently observed glycoprotein in renal transplant recipients with HCMV infections in Kuwait27 and also among children living at the Phayathai Babies' Home in Nonthaburi Province, Thailand.17 gB1 was also predominant in a study of immunocompromised patients undergoing organ transplantation29 or with congenital infection in a Chinese population.30
A recent study evaluated the prevalence of the gB genotype of HCMV in UC patients in Iran and reported that gB1 was the most prevalent genotype in UC patients.31 In our study, gB1 was successfully amplified in only 2 of 3 samples (66.7%) from UC patients, followed by gB3 (1/3, 33.3%). Although a very small number of samples were genotyped for gB, our findings confirm that gB1 gene is predominant genotype in UC patients. In our study, the colectomy rate was 33.3% in gB1 gene positive patients despite treated with ganciclovir that revealed that patients with gB1 gene of HCMV are associated with surgical intervention and/or fatal outcomes. However, our results did not reach statistical significance (data not shown), which might be related to the small case number. Hence, out results agree with previously reported study whether the association of the gB1 gene with invasive disease in solid organ transplantation was reported.32 Very recently, 1 study showed the negative association of gB1 gene of HCMV with clinical presentation.33
With respect to the gN genotype of CMV, only gN3 was detected among UC patients in the present study. Among the 4 successfully amplified gN3 samples, gN3a and gN3b were distributed equally, and all of these patients were treated with corticosteroids (oral). These results are similar to those of a previous study in which gN3a was the most frequently observed genotype in Chinese hematopoietic stem cell transplantation patients.34 The gN3b genotype was reported as the most prevalent genotype in a study of patients with inflammatory breast cancer.35 However, gN3 was not found to be frequently associated with other populations of immunocompromised patients. By contrast, gN1 has been frequently observed among solid organ transplant patients and acquired immunodeficiency syndrome patients. Whereas gN2 is commonly observed in solid organ transplant patients, gN3 is common among lung transplant recipients.363738
The present study is the first to report gN genotyping of cytomegalovirus in UC, and gN3, particularly gN3a and gN3b, was identified as the most frequently observed gN genotype. Among the gN3 positive patients, 25% of patients (1/4) with gN3b received antiviral therapy and colitis symptoms improved. The other patients with gN3b (25%, 1/4) and gN3a (50%, 2/4) did not receive ganciclovir and entered into remission. Our results suggest that gN3a and gN3b genotypes of HCMV are associated with less virulence in UC patients. The previous studies also have demonstrated that gN3a represent a less virulent genotype compared with other gN genotypes.39 However, geographic influences have also been implicated as a cause of the observed variation in gN genotypes. A previous study suggested that the gN2 genotype is widely distributed in North America and Europe but is not prevalent in Asian countries and Australia.40 Our results confirm this suggestion as the gN2 genotype was not amplified from any of the samples.
With respect to the gH genotype of cytomegalovirus, our study suggests that both gH1 and gH2 genotypes can affect UC active patients. However, the frequencies of gH2 (75%) and gH1 (12.5%) differed significantly (P<0.05) in UC active patients in our study, suggesting an association with UC activity. Our study confirms the results of previous 2 studies where gH2 variant is the most prevalent genotype in congenital CMV infection41 and in infants with thrombocytopenic purpura.42 By contrast, 1 study has reported that gH1 is more frequently observed in a Japanese population of infants infected congenitally.43 Another study reported that gH2 was more frequently observed (60% and 67%, respectively) than gH1 HCMV-infected newborns and infants in Poland. It has further been suggested that the gH1 genotype is more virulent than gH2.44 In our study, we compared the frequency of distribution of gH genotypes among UC active patients with HCMV receiving immunosuppressive drugs. The gH2 variant of HCMV was significantly more prevalent than the gH1 variant in UC active patients receiving immunosuppressive drugs (P<0.05), possibly indicating weaker virulence. Among the gH2 positive 75% of patients, 62.5% of patients (5/8) and 12.5 % of patients (1/8) were experienced severe and moderate symptoms, respectively. However, 25% (2/8) had mild colitis endoscopically. Out of these 75% of gH2 gene positive patients, 33.3% of patients received ganciclovir. Among these, antiviral therapy improved remission and surgery-free survival outcomes in 16.7% of patients and other 16.7% of patients underwent surgery under ganciclovir therapy along with steroid, anti-tumor necrosis factor. However, symptoms were improved in the other 50% of gH2 gene and 12.5% of gH1 positive patients without antiviral. Although very small number of patients were analyzed, our study suggested that gH1 gene is less virulent in UC patients whether follow-up should be done carefully gH2 gene positive UC patients since 16.7% needed surgery and/or ganciclovir.
One UC active patient (12.5%) in our cohort exhibited mixed infection of gH variants. Several studies have shown that immunocompromised patients acquire mixed infections of HCMV genotypes. Furthermore, infection with mixed genotypes has been associated with increased morbidity45 in infected individuals.46 In our study, only 1 patient experienced mixed infections of gH genotype (gH1 and gH2 both), and symptoms were improved without antiviral in spite of having severe left-sided colitis (Mayo score, 9) for 26 years. However, the number of samples in our study was very small, and the results should be considered with caution.
There are other limitations in our study. First, PCR was not successful in 3 of the 11 HCMV-positive samples in this present study. Despite repeated efforts, the PCR results remained negative. The inability to amplify UL55, UL73, and UL75 using genotype-specific PCR primers in some samples may have been due to a low concentration or degradation of HCMV DNA or the presence of base pair mutations preventing primer binding and elongation. Second, the sequencing of gB and gN was performed on PCR-amplified samples; consequently, the results only reflect the major genotype detected, and mixed infections were not addressed. Nevertheless, these results provide a preliminary report on the prevalence of the gB, gN, and gH genotypes in an Asian population of UC patients with HCMV.
In conclusion, our results demonstrated that the gB1, gN3, and gH2 genes of HCMV are the most frequently observed genotypes in UC active patients. Our study also reported that gN3 gene is less pathogenic in UC patients. On the other hand, gB1 and gH2 gene are pathogenic in UC patients and gB1 gene positive patients have more fatal outcomes. Antiviral treatment is recommended in gB1 gene positive UC patients to improve outcome. Follow-up should be done carefully in gH2 gene positive UC patients and antiviral should be prescribed as necessary. The number of UC active patients with HCMV infection analyzed in our study was very small, and thus we cannot emphasize the differences in genotype distribution. However, there have been no similar molecular epidemiological studies of HCMV infection in UC active patients among the Asian population. If specific genotypes do have a higher prevalence in UC patients, this knowledge would be helpful in predicting the severity of UC based on HCMV analysis and developing preventive measures. Further studies using a larger group of UC patients with HCMV infection are necessary to confirm the obtained results and to determine the clinical significance of our findings.
ACKNOWLEDGEMENT
The authors are grateful to clinical staff in the Department of Infectious, Respiratory, and Digestive Medicine and the Department of Endoscopy at the University of the Ryukyus.
Notes
FINANCIAL SUPPORT: The authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICT OF INTEREST: No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION: S.N. and A.H. were involved in conception and design of the study. S.N., A.H., A.I., T.O., Tetsu Kinjo, T.H., Takeshi Kinjo, and G.L.P. were involved in acquisition of data. S.N. and A.H. were involved in drafting of the manuscript. J.F. was involved in supervising the project. All authors were involved in interpretation of data and critical revision of the manuscript and have approved the final manuscript for submission.