Melatonin in the colon modulates intestinal microbiota in response to stress and sleep deprivation
Article information
Abstract
Background/Aims
Stress is closely related to the deterioration of digestive disease. Melatonin has potent anti-inflammatory properties. The objective of this study was to determine the effect of water stress (WS) and sleep deprivation (SD) on intestinal microbiota and roles of melatonin in stressful condition.
Methods
We used C57BL/6 mice and specially designed water bath for stress and SD for 10 days. We measured melatonin concentrations in serum, feces, and colon tissues by high-performance liquid chromatography. Genomic DNA was extracted from feces and amplified using primers targeting V3 to V4 regions of bacterial 16S ribosomal RNA genes.
Results
Compared to the control, melatonin concentration was lower in the WS and SD. Fecal concentration was 0.132 pg/mL in control, 0.062 pg/mL in WS, and 0.068 pg/mL in SD. In colon tissue, it was 0.45 pg/mL in control, 0.007 pg/mL in WS, and 0.03 pg/mL in SD. After melatonin treatment, melatonin concentrations in feces and colon tissue were recovered to the level of control. Metagenomic analysis of microbiota showed abundance in colitogenic microbiota in WS and SD. Melatonin injection attenuated this harmful effect. WS and SD showed decreased Lactobacillales and increased Erysipelotrichales and Enterobacteriales. Melatonin treatment increased Akkermansia muciniphila and Lactobacillus and decreased Bacteroides massiliensis and Erysipelotrichaceae.
Conclusions
This study showed that stress and SD could affect intestinal dysbiosis and increase colitogenic microbiota, which could contribute to the aggravating digestive disease. Melatonin concentrations in feces and colon tissue decreased under WS and SD. Melatonin treatment brought recovery of melatonin concentration in colon tissue and modulating dysbiosis of intestinal microbiota.
INTRODUCTION
Inflammatory bowel disease (IBD) is caused by a combination of environmental factors, intrinsic factors, and intestinal microbiota in patients with genetic predisposition, resulting in abnormal immune regulation and ultimately chronic intestinal inflammation [1]. Persons with a genetic predisposition to IBD are more likely to develop IBD and patients in remission state may deteriorate when exposed to severe stress and chronic sleep disturbances related to examination, admission, bereavement, and divorce [2]. These results suggest that chronic sleep disturbances may break the immune balance in the intestine, induce inflammation, and lead to the development of IBD. In fact, some studies have reported that patients with IBD have a high frequency of sleep disorders and low sleep quality [3,4]. However, the mechanism by which stress and sleep disorders directly affect IBD remains unclear. Dysbiosis of intestinal microbiota has been suggested as a substantial cause of IBD [5]. It is assumed that a group of microbial populations beneficial to humans are lacking and an abnormal immune response of intestinal epithelial cells to intestinal microbiota may cause chronic inflammation [5]. As we know, intestinal hormones play an important role in maintaining normal immune responses of intestinal epithelial cells to intestinal microbiota. It is suspected that stress and sleep disturbances can affect intestinal hormones and cause chronic changes in intestinal microbiota, triggering to IBD in vulnerable patients.
Melatonin (N-acetyl-5-methoxytryptamine) has been used for sleep control. It is also a neurotransmitter among intestinal hormones. Many previous reports have shown that melatonin has operative anti-inflammatory effects in experimental colitis [6-8]. Recently, it has been reported that melatonin can modulate specific gut microbiota activity and abundances [9]. It has been shown that melatonin can influence the swarming and motility of human intestinal bacteria, especially Enterobacter aerogenes [9]. Melatonin can also prevent obesity by modulating gut microbiota with a decrease in the ratio of Firmicutes to Bacteroidetes but an increase in the abundances of Akkermansia in high fat diet (HFD)-fed mice [10]. However, it remains unknown whether melatonin can regulate intestinal microbiota in response to stress and sleep deprivation. Therefore, the purpose of this study was to determine the effect of stress and sleep deprivation on intestinal microbiota and roles of melatonin using mouse model.
METHODS
1. Animals and Experimental Design
We used C57BL/6 mice and specially designed water bath for stress and partial sleep deprivation (for 6 hours during light cycle) as described previously [8]. Male C57BL/6 mice at age 9 weeks weighing 25–30 g, 6 mice in each group (Orient, Seongnam, Korea) were used after obtaining animal experiment ethics committee approval. They were kept at 22°C to 24°C, humidity of 55% to 60%, and 12-hour light/12-hour dark. They were provided free access to water and food. These mice were housed for 10 days in a specially designed water bath for stress and sleep deprivation. We used specially designed water bath with multiple platforms [7]. The water bath had either wide platform for normal sleep or narrow platform for sleep deprivation, and it was filled with water 4 cm from the base. The water in the cage was freshly replaced daily. The experiment was divided into normal sleeping group and sleep deprivation group from 2 PM to next day 10 AM by differentiating the radius of the platform in which the mouse was sitting in the water bath. Melatonin at a dose of 10 mg/kg or normal saline for placebo was administered to the mice intraperitoneally for 10 days (Fig. 1). Body weight of each mouse was measured daily. We planned euthanasia using CO2 inhalation in mouse which showed more than 20% loss from initial weight. All mice were sacrificed on 11th day using inhalation anesthesia. We used anesthetic at a concentration of 2.5% to 3% with a mixture of oxygen (55 psi & 500 mL/min) and isoflurane. Total blood was obtained from abdominal vena cava puncture soon after anesthesia. And then stool samples and colon tissues were obtained. This animal study was approved by the Institutional Animal Care and Use Committee of Eulji University Nowon Eulji University Hospital (EUIACUC 16-19).

Schematic diagram of the experiment design. Group I (n=6): control. Group II (n=6): water stress: these mice were housed in a specially designed water bath for stress for 10 days. Group III (n=6): water stress+melatonin, melatonin at a dose of 10 mg/kg was administered to these mice intraperitoneally for 10 days. Group IV (n=6): water stress+sleep deprivation. The experiment was divided into normal sleep group and sleep deprivation group by differentiating the radius of the plat form in which the mouse was sitting in the water bath. Group V (n=6): water stress+sleep deprivation+melatonin.
2. Measurement of Melatonin Concentration
On the 11th day, all mice were sacrificed and melatonin levels in blood, feces, and colon tissues of these mice were measured. We measured melatonin concentration by high-performance liquid chromatography using acetyl-D3 melatonin as an internal standard due to presence of very small amount only. A rapid method for the determination of melatonin in mouse has been developed in our lab using liquid chromatography coupled with tandem mass spectrometry with positive electrospray ionization. The preparation of serum sample was established by using liquid-liquid extraction with acetonitrile while the preparation of fecal and tissue samples was established by using solid-liquid extraction with dichloromethane.
3. Gut microbiota Analysis by Metagenomic Analysis
At the end of sleep deprivation, mice were sacrificed and feces were collected. Feces samples were stored at –80°C until DNA extraction and measurement of intestinal microflora under the same conditions.
Genomic DNA was extracted from feces and amplified using primers targeting V3 to V4 regions of bacterial 16S ribosomal RNA (rRNA) genes. Sequencing was performed at ChunLab, Inc. (Seoul, Korea) with Illumina MiSeq Sequencing system (Illumina, San Diego, CA, USA) according to the manufacturer’s instructions.
1) PCR Amplification and Illumina Sequencing
PCR amplification was performed using primers targeting V3 to V4 regions of the 16S rRNA gene with extracted DNA as template. Primers of 341F (5’-TCGTCGGCAGCGTCAGATGTGTATAAGAGACAG-CCTACGGGNGGCWGCAG-3’) and 805R (5’-GTCTCGTGGGCTCGG-AGATGTGTATAAGAGACAGGACTACHVGGGTATCTAATCC-3’) were used to amplify bacteria. PCR amplifications were conducted under the following conditions: initial denaturation at 95°C for 3 minutes, followed by 25 cycles of denaturation at 95°C for 30 seconds, primer annealing at 55°C for 30 seconds, and extension at 72°C for 30 seconds, and a final elongation step at 72°C for 5 minutes. Secondary amplification for attaching Illumina Nextera barcode was performed with i5 forward primer (5’-AATGATACGGCGACCACCGAGATCTACAC-XXXXXXXXTCGTCGGCAGCGTC-3’; X indicates the barcode region) and i7 reverse primer (5’CAAGCAGAAGACGGCATACGAGATXXXXXXXX-AGTCTCGTGGGCTCGG-3’). The condition of secondary amplification was the same as the previous one, except that the amplification cycle set to be 8. PCR products were confirmed by 2% agarose gel electrophoresis and visualized under a Gel Doc system (Bio-Rad, Hercules, CA, USA). Amplified products were purified with QIAquick PCR purification kit (Qiagen, Valencia, CA, USA). Equal concentrations of purified products were pooled together and non-target products were removed with Ampure beads (Agencourt Bioscience, Beverly, MA, USA). The quality and product size were assessed on a Bioanalyzer 2100 (Agilent, Palo Alto, CA, USA) using a DNA 7500 chip. Mixed amplicons were pooled and sequencing was conducted at ChunLab, Inc. with Illumina MiSeq Sequencing system according to the manufacturer’s instructions.
2) MiSeq Pipeline Method
Processing of raw reads started with quality check and filtering of low quality (< Q25) reads using Trimmomatic version 0.321. After quality control pass, paired-end sequence data were merged using PandaSeq2. And then primers were trimmed using ChunLab’s in-house program at a similarity cutoff value of 0.8. Sequences were then denoised using Mothur’s 3 pre-clustering program to merge sequences and extract unique sequences while allowing up to two differences between sequences.
3) Bioinformatic Analysis
EzTaxon database was used for taxonomic assignment using BLAST 2.2.22. Pairwise alignment was used to calculate similarity. Uchime and non-chimeric 16S rRNA database from EzTaxon were used to detect chimera on reads that contained less than 97% best hit similarity rate. Sequence data were then clustered using CD-Hit and UCLUST. For analyzing alpha diversity, we rarified the operational taxonomic unit (OTU) to several metrics, including curves of OTU rank, rarefaction, and Shannon. We then calculated indices of Shannon, Chao1, Simpson, and ACE. For beta-diversity analysis, heatmap of RDA-identified key OTUs, principal component analysis, principal coordinate analysis, nonmetric multidimensional scaling, and unweighted pair group method with arithmetic mean were performed using QIIME. Linear discriminant analysis (LDA) effect size (LEfSe) analysis was performed for quantitative analysis of biomarkers among each group [11]. Briefly, LEfSe analysis, LDA threshold of > 4, used nonparametric factorial Kruskal-Wallis sum-rank test. Then unpaired Wilcoxon rank-sum test was used to identify the most differently abundant taxa.
4. Statistical Analysis
All statistical analyses were done using SPSS version 20 (IBM Corp., Armonk, NY, USA). Differences between groups were statistically analyzed using Kruskal-Wallis test. Statistical significance was considered at P<0.05.
RESULTS
1. Weight Change
The water stress group and the sleep deprivation group presented significant weight loss compared to the control group. Melatonin administration did not reduce weight loss in that stressful condition (Fig. 2A).

Animal experimental results. (A) Changes in body weight in each group. Stress induced significant weight loss without intestinal histologic change. No significant reduction in weight loss during water stress was observed even though melatonin administration. (B) Colon length in each group. (C) Histological analyses of colon (H&E, ×100). There were no significant changes in colon length or microscopic inflammatory change. WS, water stress; SD, sleep deprivation; M, melatonin.
2. Histologic Analysis
There were no significant changes in colon length or microscopic inflammation among groups: 7.65 ± 1.0 cm in the control group, 7.60 ± 0.79 cm in the water stress group, 7.57 ± 0.74 cm in the water stress with melatonin group, 7.52 ± 0.72 cm in the water stress with sleep deprivation group, and 7.51 ± 1.0 cm in the sleep deprivation with melatonin group (Fig. 2B and C).
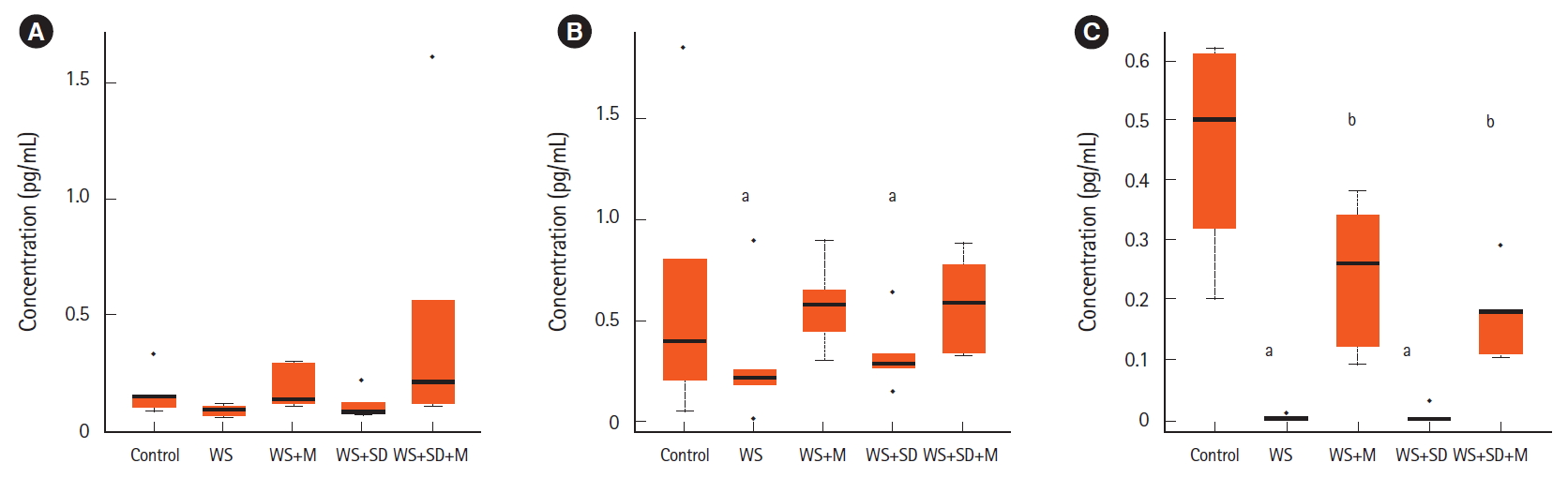
3. Measurement of Melatonin Concentration
Serum concentrations of melatonin were not significantly changed between water stresses (0.09 pg/mL) or sleep deprivation group (0.11 pg/mL) compared to the control group (0.16 pg/mL). They were increased by melatonin administration (0.18 and 0.48 pg/mL). In feces, water stress group and sleep deprivation group showed significantly lower concentrations of melatonin than the control group (0.062, 0.068, and 0.132 pg/mL, P<0.05). When melatonin was administered, the level of melatonin in feces was well maintained during water stress and sleep deprivation (0.116 and 0.118 pg/mL). In colon tissues, concentrations of melatonin in water stress and sleep deprivation groups were very low (0.007 and 0.03 pg/mL) compared to the control group (0.45 pg/mL, P<0.05). The level of melatonin in the colonic tissue had decreased less after melatonin administration (0.24 and 0.17 pg/mL, P<0.05) (Fig. 3).
4. Changes in Microbiota under Water Stress and Sleep Deprivation
Metagenomic analysis of intestinal microbiota showed significant shifting of abundance after water stress and/or sleep deprivation. In the water stress and water stress with sleep deprivation group, the abundance of Lactobacillales was significantly decreased, but there was increased abundance in colitogenic microbiota such as Enterobacteriales and Erysipelotrichales at order level compared to control group (Fig. 4A). Richness, Shannon index, or Simpson index showed no statistically significant changes among groups (Fig. 4B). At phylum level, it showed increased abundance of Proteobacteria and Bacteroidetes in water stress and water stress with sleep deprivation group (Fig. 4C, inner circle: phylum; outer circle: species).
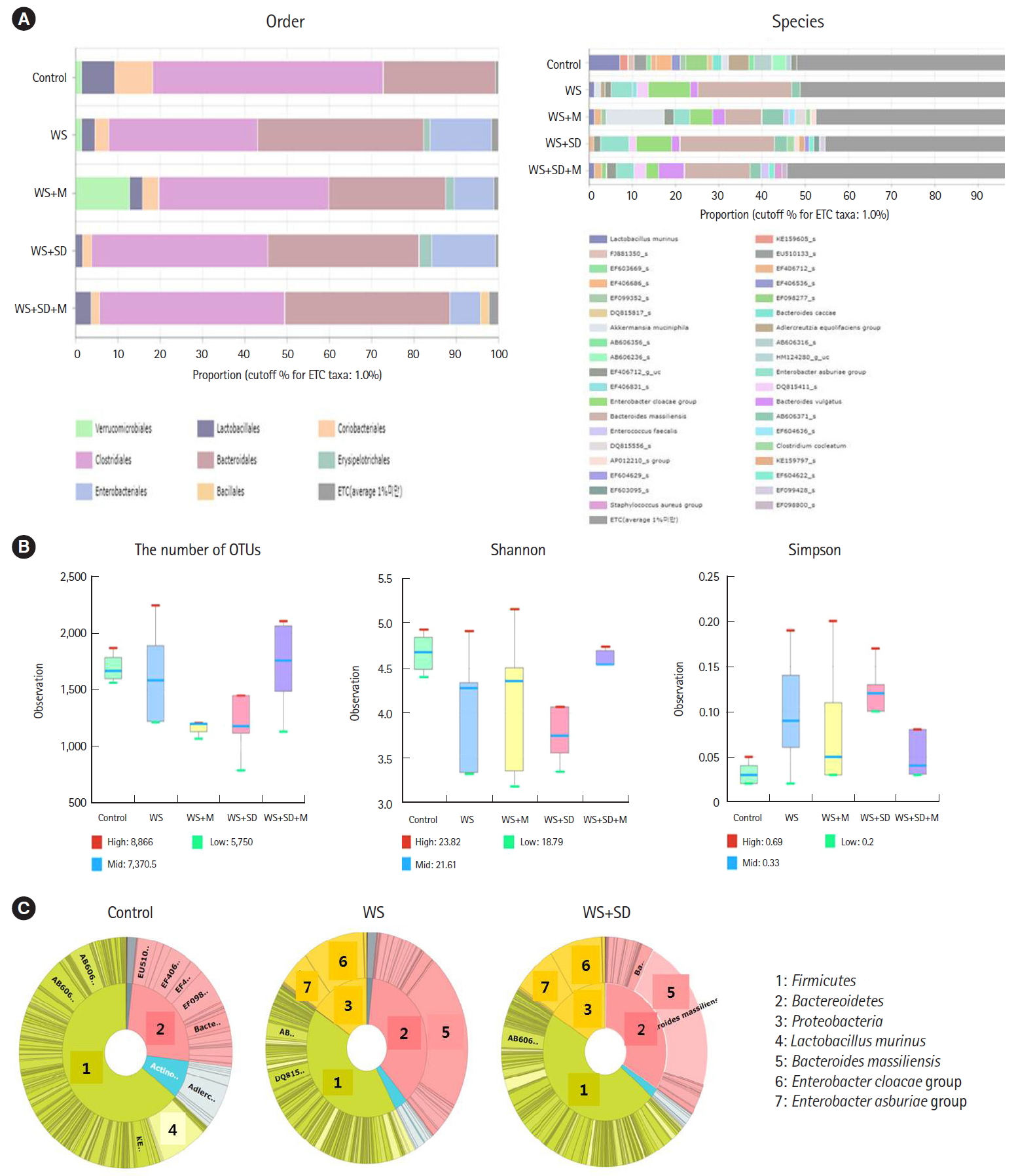
Analysis of intestinal microbiota in each group. (A) In water stress and/or sleep deprivation, there were significantly increased abundance in order of colitogenic microbiota such as Enterobacteriales and Erysipelotrichales. (B) No significant change in the Richness, Shannon index, Simpson index, or the number of operational taxonomic units (OTUs) among groups. (C) Increased abundance of Proteobacteria and Bacteroidetes at phylum level in water stress and/or sleep deprivation group (inner circle: phylum; outer circle: species). WS, weight loss; SD, sleep deprivation; M, melatonin.
To compare abundant bacterial species associated with water stress and sleep deprivation, we applied the LEfSe method (Fig. 5). The results showed that the relative abundant bacteria in the control group were Lactobacillus murinus, Adlercreutzia equolifaciens group, Bacteroides caccae, Akkermansia muciniphila. Comparing to control in the water stress and sleep deprivation group, Bacteroides massiliensis, Enterobacter cloacae group, Enterobacter asburiae group, Bacteroides vulgatus were relatively abundant.
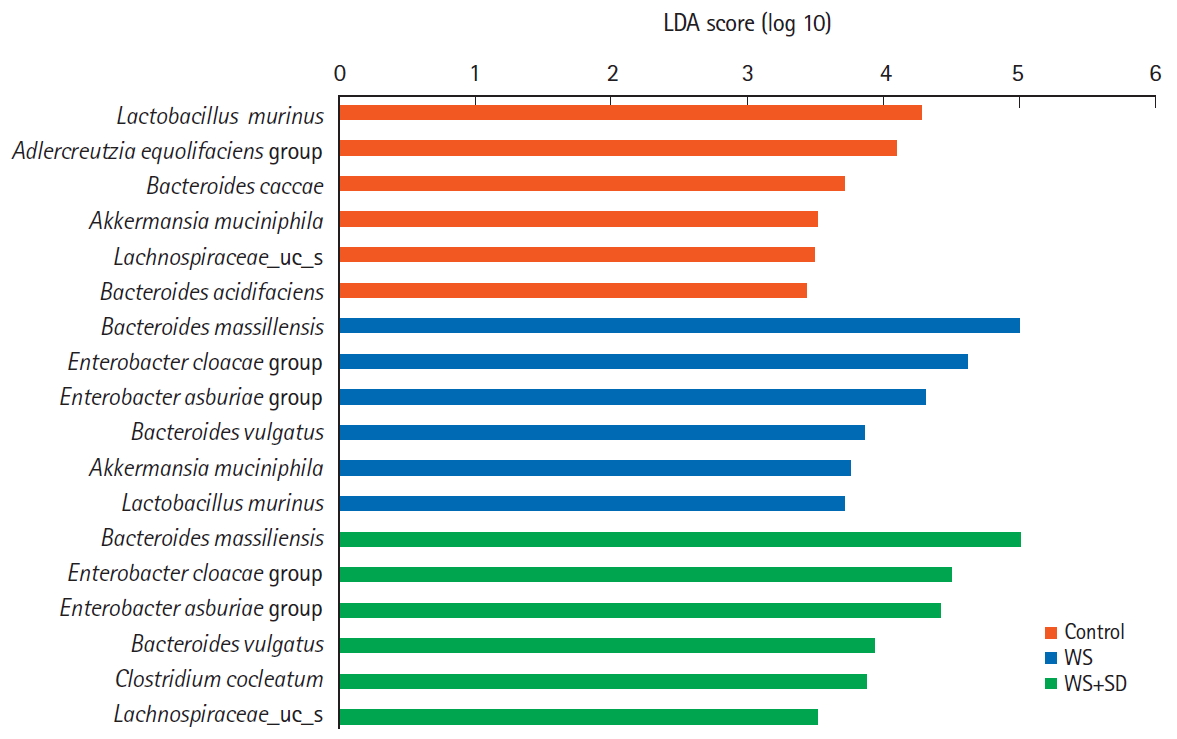
Compared relative abundant species in intestinal microbiota among each group. LDA, linear discriminant analysis; WS, water stress; SD, sleep deprivation.
At species level, water stress and/or sleep deprivation group showed dramatic changes, such as marked decrease in the abundance of L. murinus, but significant increase in the abundance of B. massiliensis. E. asburiae, Clostridium cocleatum (Fig. 6, P<0.05 by Kruskal-Wallis H test).
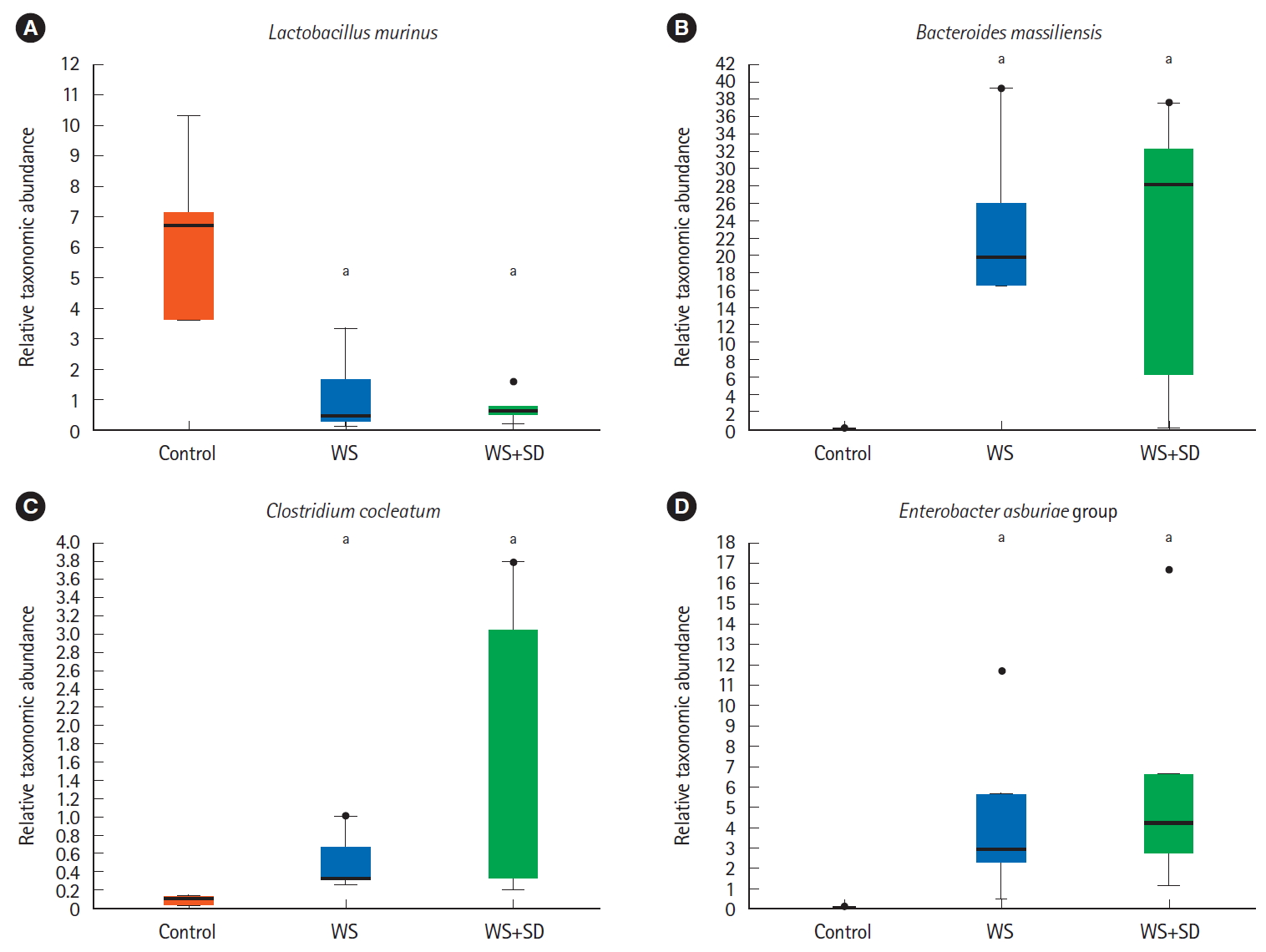
(A-D) At species level, water stress (WS) with sleep deprivation (SD) group showed significant change such as decreased abundance of Lactobacillus murinus and increased abundance of Bacteroides massiliensis. Clostridium cocleatum group, Enterobacter asburiae group (P<0.05 by Kruskal-Wallis H test). aP<0.05.
5. Effect of Melatonin on Intestinal Microbiota
The Richness, Shannon index, or Simpson index showed no statistically significant changes after melatonin treatment (Fig. 4B). Administration of melatonin attenuated the microbial changes in water stress and sleep deprivation. The decrease in the abundance of Lactobacillus has been reduced (Fig. 7A, P<0.05 by Kruskal-Wallis H test). Some mice showed significant increase in the abundance of A. muciniphila after melatonin administration. The abundance of B. massiliensis, in water stress and water stress and sleep deprivation had been reduced after melatonin treatment. Erysipelotrichaceae had been reduced only in the water stress and sleep deprivation with the melatonin group (Fig. 7B, P<0.05 by Kruskal-Wallis H test).
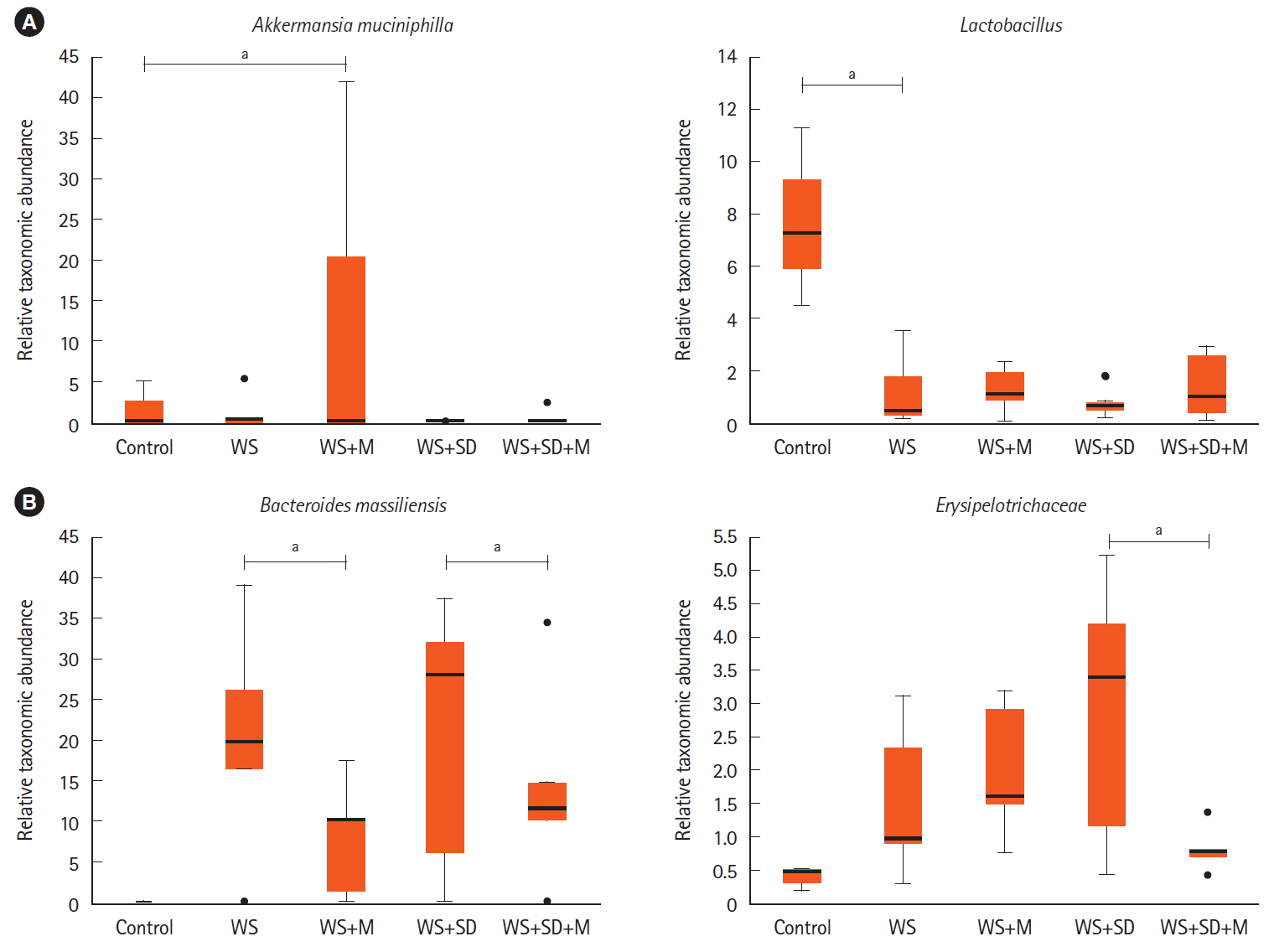
Changes in intestinal microbiota after melatonin treatment. (A, B) Administration of melatonin resulted in changes such as increased abundance of Akkermansia muciniphila and Lactobacillus but decreased abundance of Bacteroides massiliensis, and Erysipelotrichaceae (P<0.05 by Kruskal-Wallis H test). aP<0.05. WS, water stress; M, melatonin; SD, sleep deprivation.
DISCUSSION
In this study, we observed that melatonin concentrations were reduced in stool and colon tissues under stressful environment without histologic change. Through gut microbiota profiling, our results suggest that stress and sleep deprivation could modulate intestinal microbial environment by increasing colitogenic microbiota. Melatonin treatment attenuated these dysbiotic changes and reduced the decrease of the abundance of Lactobacillus and Akkermansia compared to control.
Melatonin is synthesized from tryptophan. It can modulate a variety of molecular pathways including sleep, circadian rhythms, immune function, apoptosis, proliferation, and oxidative stress [11-13]. Melatonin influences physiological functions of the GI tract, including permeability [14,15], bicarbonate secretion,15 energy utilization [16], motility [17], and tight junction [17] of intestinal epithelium. It also attenuates several GI diseases, including IBS, IBD, necrotizing enterocolitis, and ischemic injuries [18,19]. Unfortunately, very small quantities are present in the GI tract, so measurement of the concentration of melatonin is difficult. To measure the small quantity of melatonin, we developed a rapid method using liquid chromatography coupled with tandem mass spectrometry with positive electrospray ionization.
In this study, serum levels of melatonin were not significantly different between the control group and the water stress or sleep deprivation group. However, in feces and colon tissues, concentrations of melatonin were significantly decreased in the water stress or sleep deprivation group compared to the control group. After melatonin administration, concentrations of melatonin in the water stress and sleep deprivation group were improved similar to the control group. Our results suggest that serum melatonin is mainly produced from the pineal gland [16] and was not significantly affected by stressful conditions. In contrast, melatonin in the GI tract is generated from the intestinal enterochromaffin cell and is significantly decreased in water stress and sleep deprivation.
Melatonin has potent anti-inflammatory effects. So, it is possible that low melatonin concentration in the GI tract could reduce an intestinal resilience to inflammation. This is supported by our previous report that melatonin treatment attenuate dextran sodium sulfate induced colitis with sleep deprivation [6]. The recovery of these changes after melatonin administration suggests that melatonin treatment have a role in the recovery of inflammatory bowel condition.
It has been suggested that stress can play a detrimental role in IBD by increasing intestinal permeability and facilitating intestinal translocation of bacteria which in turn can stimulate innate and adaptive immune responses [20]. However, there are limited data suggesting that stress directly exacerbates intestinal inflammation [20]. In our study, water stress and sleep deprivation resulted in significant decrease in body weight of mice compared to the control group without water stress or sleep deprivation. Administration of melatonin did not help maintaining body weight. However, histological examination revealed no inflammatory reaction of the intestine in all groups. Thus, weight loss was not related to histological changes such as intestinal inflammation or ulcer due to stress. Our results also suggest that stress does not directly induce histological inflammatory response in the intestine.
However, in a study on poor sleep and inflammatory markers in adolescent cohorts, insomnia and objective shortening of sleep group have shown higher CRP level and other inflammatory markers [21]. Meta-analysis of adults has also shown that sleep disturbance is associated with elevated CRP, suggesting that sleep disturbance is associated with systemic inflammatory responses [22,23]. Also, IBD patients have inferior sleep quality, significantly prolonged sleep latency, and frequent sleep fragmentation. Patients who have experienced sleep disturbances even in the remission period are more likely to have recurrence and chronic inflammatory status [23,24]. These results support a possible explanation that stress and sleep disturbance induce both lowering melatonin level and inducing systemic inflammatory response, and then deteriorating IBD.
On microbiota analysis, it showed the significant changes of intestinal microbiota induced by water stress and sleep deprivation. Benefits of intestinal microbiota to the host’s physiology include nutrition [25], immune development [26], and host defense [27].
The intestinal microbiota supplies energy and nutrients to the host. It provides short-chain fatty acids by fermenting resistant starch or dietary fiber. It plays an important role in the development of host’s immune system. The host’s immune system mutually affects the structure and function of the intestinal microbiota. The intestinal microbiota also contributes to host defense against pathogens [27]. Germ-free animals are vulnerable to infection by intestinal pathogens.
In our study, the decrease in the abundance of Lactobacillus and A. muciniphila [28,29] which are known as beneficial bacteria in intestines and the increase in the abundance of B. massiliensis [30] and C. cocleatum [31,32] known to cause inflammation can explain another possible mechanism that stress affects the deterioration of IBD. These changes after water stress and sleep deprivation were suppressed by simultaneous administration of melatonin. In the melatonin treated group, there was significant increase of A. muciniphila, a beneficial gut microbiota that could reduce inflammation. After the administration of melatonin, colitogenic bacteria such as E. cloacae [33], E. asburiae [34], B. massiliensis, and Erysipelotrichaceae were also decreased. Results of this study are consistent with previous melatonin studies. It has been shown that melatonin can reduce inflammatory response through TLR4 (Toll-like receptor 4)-associated signaling pathways. TLR4 is an important receptor for intestinal microbial response receptor, suggesting that melatonin can directly affect the intestinal microbiota [35]. On the other hand, it has been reported that melatonin can affect the motility and activity of E. aerogenes, a specific human gut bacteria, in a cyclic manner, suggesting that melatonin can directly interact with bacteria [9]. In this study, it was shown that the administration of melatonin resulted in an increase of A. muciniphila. In a previous study using obese mouse induced by HFD, the abundance of Akkermansia is increased by administration of melatonin [10], consistent with our findings. They have reported that obesity can be prevented by changing bacterial compositions [10]. Melatonin can also prevent obesity by modulating intestinal microbiota with a decrease in the ratio of Firmicutes to Bacteroidetes and an increase in the abundance of Akkermansia in HFD-fed mice [10].
Recently many studies showed that A. muciniphila contribute to host mucosal homeostasis. A. muciniphila colonization increases mucin production, improve barrier function and butyrate production. Additionally, it is inversely associated with intestinal inflammation and protect the epithelium from colitis by secreting extracellular vesicles [36-38].
In our study, Lactobacillus was reduced in the water stress group but was increased in the melatonin-treated group, consistent with results of melatonin administration in weaning stress mice [39]. In the melatonin treated group, B. massiliensis and Erysipelotrichaceae showed decreases in abundance. These bacteria are known to be involved in inducing IBD [39]. Another group reported that the imbalance between Erysipelotrichaceae (family) and Lachnospiraceae is one cause of IBD and the deficiency of a gene controlling the innate immunity mechanism can cause this abnormality [40]. Together, these results suggest that melatonin is related to changes in bacterial population associated with IBD.
There is increasing evidence that dysbiosis of the intestinal microbiota is associated with various human diseases, including IBD, IBS, obesity and metabolic syndrome. It has been reported that the composition of microbiota in IBD is different from that in healthy subjects [41-43]. However, a direct causal relationship between dysbiosis and IBD has not been confirmed. Despite the fact that bacterial composition in the intestine is likely to be related to food [44], a similar decrease in the relative proportion of Firmicutes has been consistently observed in Eastern and Western CD patients with completely different food cultures [45]. This suggests that the onset of the disease is associated with a situation where changes in intestinal bacterial composition are induced besides eating habits. Previous studies have reported that relative proportions of Faecalibacterium and Lachnospiraceae are decreased in CD patients, indicating that these compositional changes are important for the development of IBD [5]. In our study, the change of microbiota in water stress and sleep deprivation showing increased abundance of B. massiliensis and C. cocleatum known to be colitogenic bacteria with reduction of Lactobacillus and A. muciniphila known to be beneficial bacteria, similar to intestinal bacterial composition of IBD patients. This is a significant finding in that our microbiota study suggests change of intestinal bacteria has already begun before inflammation has occurred. Also, changes in compositions can be alleviated by administration of melatonin. The abundance of beneficial bacteria indicates the importance of melatonin as a helpful modulator of intestinal microbiota.
Our study has limitation of, weight loss can affect microbiota composition during stressful condition. So further study is necessary in this condition.
In conclusion, this study showed that stress and sleep deprivation could affect intestinal dysbiosis and increase colitogenic microbiota, which could contribute to the aggravating digestive disease. Melatonin concentrations in feces and colon tissue were decreased under water stress and sleep deprivation. Melatonin treatment brought recovery of melatonin concentration in colon tissue and modulation of dysbiosis of intestinal microbiota.
Notes
FINANCIAL SUPPORT
This work was supported by a grant of the National Research Foundation (NRF) funded by the Ministry of Science and ICT, Republic of Korea (NRF-2016R1A2B1016426) (Park YS). They funded the all cost of animal experiments and microbiological analysis.
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTION
Design of the study: Kim SH, Park YS, Park JW, Kho Y, Jun JH. Data acquisition; data analysis and interpretation; wrote the manuscript: Kim SH, Park YS. Conceived the study; critical revision of manuscript for intellectual content: Park JW. Data acquisition: Seok PR, Choi YJ. Data analysis and interpretation: Kho Y, Jun JH, Shin JH, Jung HC, Kim EK. Approval of final manuscript: all authors.