Novel inflammatory bowel disease (IBD) specific electronic medical record allows scalable auditing of IBD severity, therapy and complications to show the current unmet need in IBD care
Article information
Inflammatory bowel disease (IBD) is a chronic condition with a relapsing-remitting course which impairs quality of life [1]. IBD requires long-term medical management resulting in significant costs to individuals and society. Annual hospitalization costs in Australia exceed 100 million Australian dollars (AUD) with overall estimated economic costs in Australia exceeding 2.7 billion AUD [2].
A 2016 audit of IBD care in Australia found a high burden of disease, inconsistent care within and across providers and poor documentation [3]. This informed the development of a dedicated independent electronic medical record (EMR) for IBD care-Crohn’s Colitis Care (CCCare). This initiative was developed and funded by an Australasian charity–Crohn’s Colitis Cure (www.c-c-cure.org), in conjunction with members of the Australia New Zealand IBD Consortium (www.ANZIBDC.com). The EMR is managed and run by the Australasian charity–CCCare (www.c-c-cure.org). The development and early usability [4], and patient acceptability [5] of CCCare have been previously described. This paper aims to document the delivery of the following objectives: (1) to implement and pilot a national, cloud-based, EMR for IBD; (2) to demonstrate this IBD-specific EMR works in routine clinical care and is able to simultaneously populate a research registry without duplicate data entry; (3) ascertain the current unmet need for more effectively delivered IBD therapy as defined by the proportion of patients in specialist care with active disease; or (4) provide an accurate picture of IBD severity, therapy, and complications in a real-world cohort of IBD patients under usual care at 2 large IBD Services.
Data were analyzed from the first 2 adult tertiary IBD centers to adopt CCCare. All patients consented to their data being entered into CCCare which included the parallel migration of non-personally identifiable information to the research registry for clinical audit and research. Health and ethics approval for the EMR to include patient data in a clinical research registry was obtained from the central Adelaide Health Ethics Research Committee (approval No. 83.20). Patient assessments and data were collected as face to face or virtual consults as part of usual clinical care. All patients from each center were included in CCCare. Data analyzed here include patient demographics, clinical assessments, medication use, hospital admission(s), investigation results such as colonoscopy and harm minimization data, e.g., vaccinations. Statistical analysis was performed using Stata version 15 (StataCorp LLC, College Station, TX, USA) and SAS 9.3 (SAS, Cary, NC, USA).
The study population included 1,785 patients, with a median age of 41.2 years (interquartile range, 30.2–55.3 years); 56.3% with Crohn’s disease (CD). The median disease duration was 10.2 years (interquartile range, 5.2–17.2 years), with disease duration over 5 years in 70.3% of those with CD and 75.6% of those with ulcerative colitis.
The study sites started using CCCare in August 2018 and June 2019. At time of writing CCCare was being used at 14 sites across Australia and New Zealand with greater than 5,000 people with IBD having care documented by CCCare. The CCCare EMR is accessible through a web interface with the software running on a cloud platform (MS Azure), ensuring all sites and users are using the same version of the EMR at any one time. Software enhancements and new features have been implemented and successfully deployed without incident over the last 2.5 years.
The EMR has been used as the prime data documentation tool in routine care for 6,669 discrete clinical assessments, 2,785 endoscopic procedures, and 6,191 calprotectin results.
At each clinical assessment, CCCare prompted recording of both physician global assessment of disease activity and patient assessment of general wellbeing. Active disease as scored by physician global assessment was present in 49.8% of clinical visits. Perhaps surprisingly, patient assessment of general wellbeing was reported as “poor” or “terrible” in 8.3% of those in clinical remission. Arthralgia was the most common extraintestinal manifestation with ongoing arthralgia in 1 of 12 patients in clinical remission. The influenza vaccination in the preceding 12 months was documented in 30.7% of those receiving a biologic or immunomodulator. Active cigarette smoking was recorded in 14% of the total cohort with 15.8% of people with CD continuing to smoke.
Disease activity during clinical visit and current medication are shown in Table 1, with those receiving anti-TNF therapy having a lower rate of active disease than other biologics (26.2% vs. 35.0% active disease, P= 0.001). Current prednisolone usage was recorded in only 2.3% of the study population, with prednisolone usage for longer than 3 months present in one-third of those on prednisolone (0.75% of total cohort).
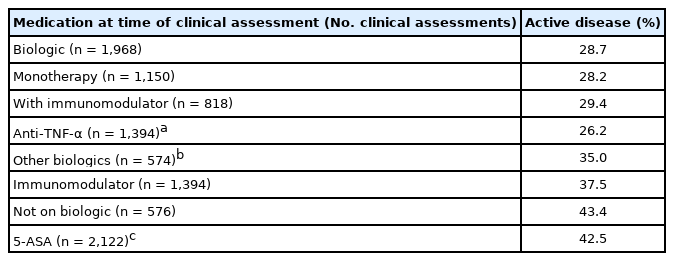
Disease Activity as per Physician Global Assessment during Each Clinical Assessment against Current Medication
Patients were assessed as having moderate to severe IBD activity in more than 1 in 10 clinical visits ( > 10%). Similarly, patients’ self-reported rating of “General Wellbeing” was poor to terrible in 1 in 10 clinical visits. There was good concordance with physician and patient assessment of IBD activity. In assessments where physicians gave a score of clinical remission the patient-reported general wellbeing was “very well” in 81.6% of encounters.
Over a 3-year period, 12.6% of the study population had IBD-related surgery recorded, with emergency surgery making up over a quarter of episodes. Stricturing disease was the most common reason for surgery (23.2%), followed by abscess (21.7%), chronic active disease (16.8%), and active perianal fistula (8.8%).
Here, a new E-tool–CCCare-seamlessly enabled auditing of IBD care and outcomes in a real-world setting. It can utilize data from 2 large IBD services, gathered during routine care, to report against the Australian IBD National Audit items from 2016 [3]. Disappointingly, the data show high rates of active disease during routine care (in 40%–50% of visits), but pleasingly, low rates of steroid use and what appears to be low rates of surgery (12.6% overall, with only 4% having emergency surgery).
CCCare can be used to examine concordance between patients’ and doctors’ judgements of disease activity. CCCare will also enable quality data on extraintestinal manifestations to be reported for incidence and prevalence and better inform their relationship to disease activity and response to various therapies in large cohorts. Already we can see the benefit in being able to reflect to clinicians the difference in likelihood of remission amongst patients on biologics compared to immunomodulatory therapy and perhaps a difference in responsivity amongst biologic classes. The impact of CCCare on clinical care requires further investigation. The value of CCCare will increase over time as the usage of the EMR spreads and the time under observation of the cohort increases.
Notes
Funding Source
This work was supported in part by a research grant from the Investigator-Initiated Studies Program of Organon (grant number: MSD IIS 55839). The opinions expressed in this paper are those of the authors and do not necessarily represent those of Organon.
Conflict of Interest
Andrews JM has been on advisory boards, been a speaker and received research and/or educational support from AbbVie, Allergan, Anatara, Bayer, Celgene, Celltrion, Ferring, Gilead, Hospira, Immunic, Janssen, MSD, Nestle, Pfizer, Sandoz, Shire, Takeda, Vifor, Novartis. Connor S has been on advisory boards, been a speaker and received research and/or educational support from AbbVie, Janssen, Pfizer, Shire, Takeda, Ferring, MSD, Vifor, Celgene, Orphan, Gilead, Celltrion, Aspen. Other authors report no conflict of interest.
Data Availability Statement
The data used in this work was generated as part of routine care at the Royal Adelaide Hospital and Liverpool Hospital. The data underlying this article will be shared on reasonable request to the corresponding author.
Author Contribution
Conceptualization: Barnes A, Walsh A, Connor S, Andrews JM. Data curation: Barnes A, Carter D, Kaazan P. Formal analysis: Barnes A. Methodology: Barnes A. Writing-original draft: Barnes A, Andrews JM. Writing - review & editing: Barnes A, Walsh A, Connor S, Andrews JM. Approval of final manuscript: all authors.
Others
The data used in this work was generated as part of routine care at the Royal Adelaide Hospital and Liverpool Hospital. The data underlying this article will be shared on reasonable request to the corresponding author.
