 |
 |
- Search
| Intest Res > Volume 21(3); 2023 > Article |
|
Abstract
Background/Aims
Methods
Results
ADDITIONAL INFORMATION
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
Ng SC has served as a speaker for Janssen, Abbvie, Takeda, Ferring, Tillotts, Menarini, Pfizer and has received research grants from Olympus, Ferring, Janssen and Abbvie. She is scientific cofounder of GenieBiome Limited and sits on the Board of Directors of GenieBiome Ltd. Hisamatsu T has performed joint research with Alfresa Pharma Co., Ltd., and EA Pharma Co., Ltd., received grant support from Mitsubishi Tanabe Pharma Corporation, EA Pharma Co., Ltd., AbbVie GK, JIMRO Co., Ltd., Zeria Pharmaceutical Co., Ltd., Daiichi-Sankyo, Kyorin Pharmaceutical Co., Ltd., Nippon Kayaku Co., Ltd., Takeda Pharmaceutical Co., Ltd., Pfizer Inc., and Mochida Pharmaceutical Co., Ltd., and received consulting and lecture fees from Mitsubishi Tanabe Pharma Corporation, AbbVie GK, Celgene K.K., EA Pharma Co., Ltd., Kyorin Pharmaceutical Co., Ltd., JIMRO Co., Janssen Pharmaceutical K.K., Mochida Pharmaceutical Co., Ltd., Takeda Pharmaceutical Co., Ltd., and Pfizer Inc. Ye BD has served on advisory boards for AbbVie Korea, Celltrion, Daewoong Pharma, Ferring Korea, Janssen Korea, Pfizer Korea, Takeda, and Takeda Korea, has received research grants from Celltrion and Pfizer Korea, has received consulting fees from BMS Pharmaceutical Korea Ltd., Chong Kun Dang Pharm., CJ Red BIO, Curacle, Daewoong Pharma, IQVIA, Kangstem Biotech, Korea Otsuka Pharm, Korea United Pharm. Inc., Medtronic Korea, NanoEntek, ORGANOIDSCIENCES LTD., and Takeda, and has received speaker fees from AbbVie Korea, Celltrion, Cornerstones Health, Curacle, Daewoong Pharma, Ferring Korea, IQVIA, Janssen Korea, Pfizer Korea, Takeda, and Takeda Korea. Except for that, no potential conflict of interest relevant to this article was reported.
Supplementary Material
Supplementary Material
Fig.┬Ā1.
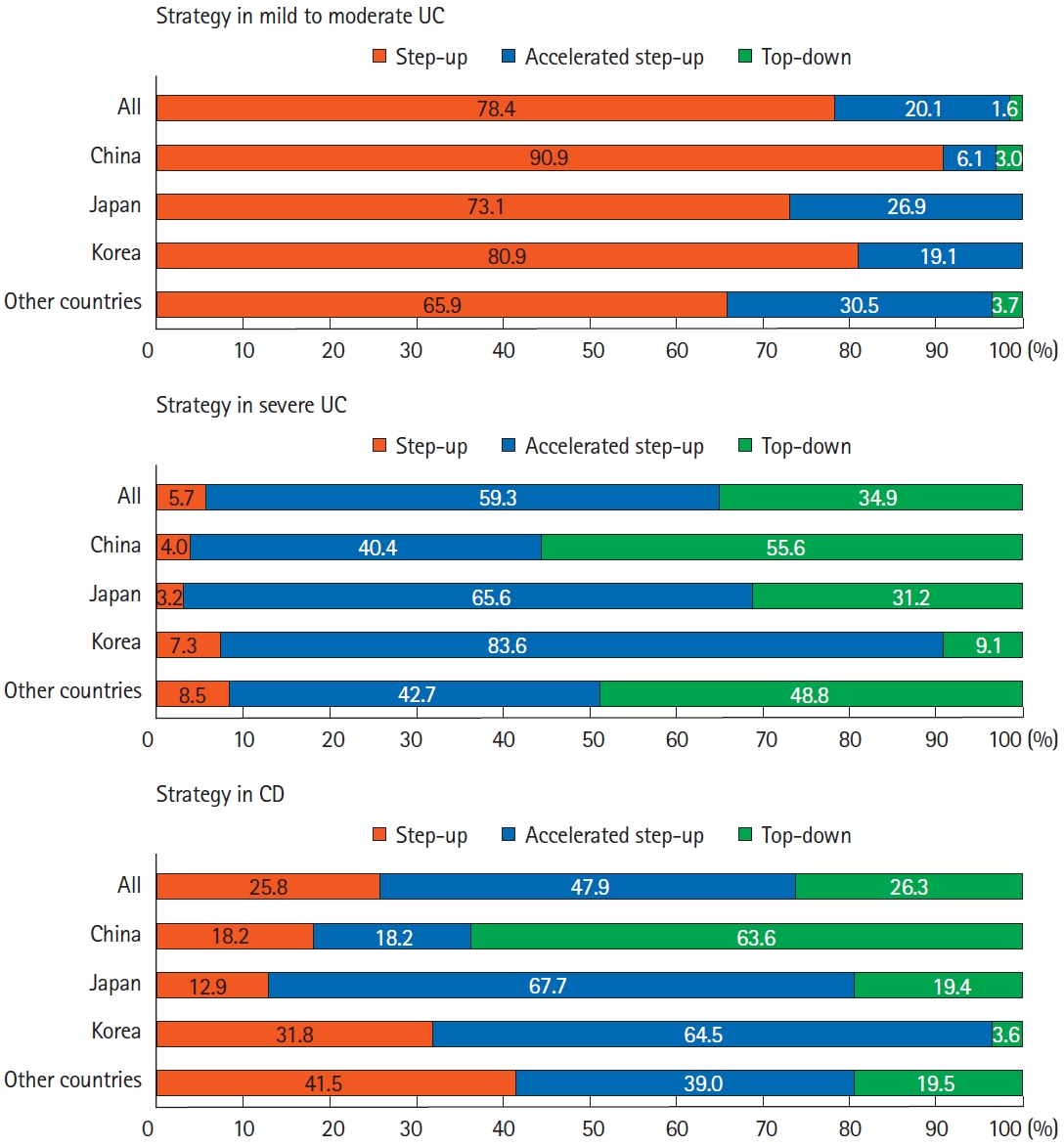
Fig.┬Ā2.
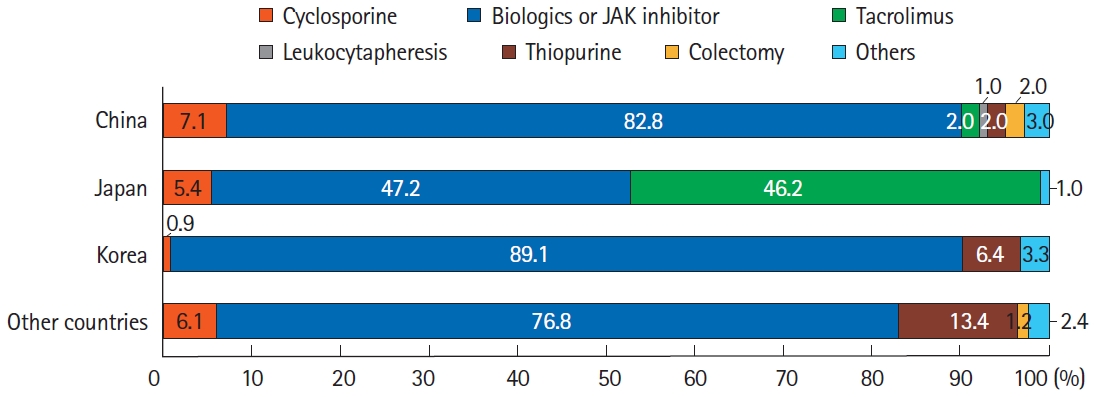
Fig.┬Ā3.
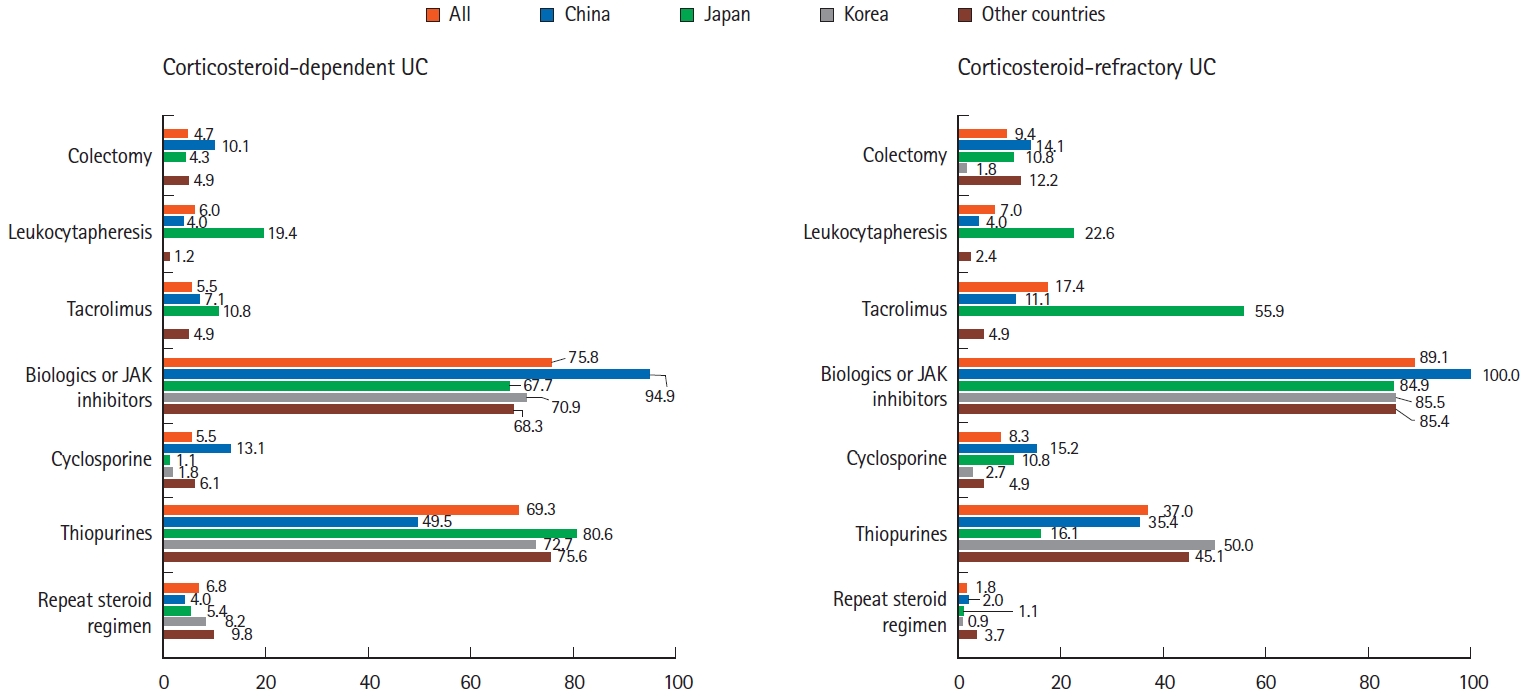
Fig.┬Ā4.

Fig.┬Ā5.
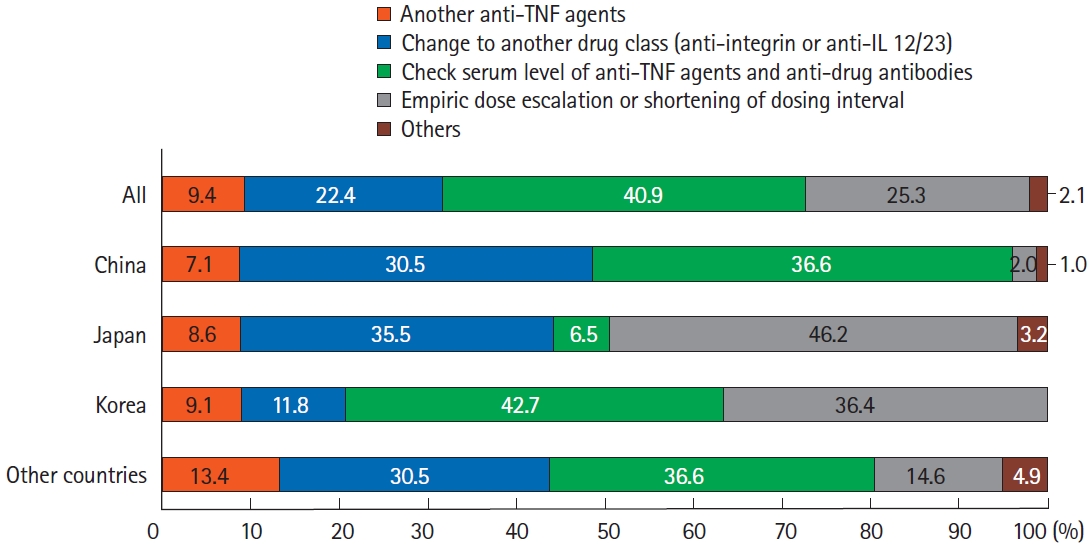
Fig.┬Ā6.
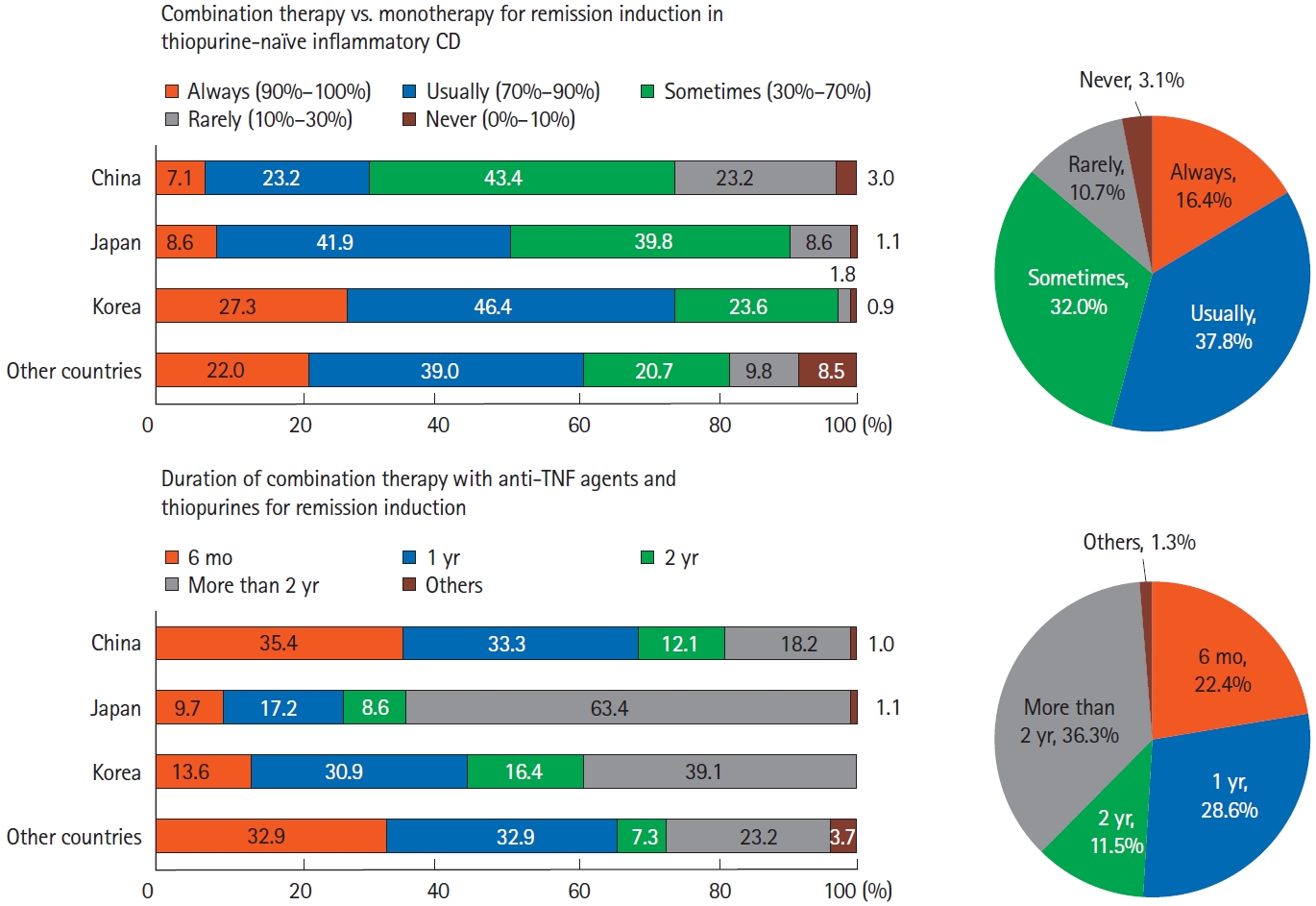
Fig.┬Ā7.

Table┬Ā1.
Table┬Ā2.
Table┬Ā3.
Values are presented as number (%). Analysis of answers to the following questions: ŌĆ£Is it available to monitor the level of 6-TGN, 6-MMP in your practice?ŌĆØ, ŌĆ£Is it available to monitor serum level of anti-TNF agents in your practice?ŌĆØ and ŌĆ£Is it available to monitor the level of serum antibodies to anti-TNF agents in your practice?ŌĆØ
TNF, tumor necrosis factor; 6-TGN, 6-thioguanine nucleotide; 6-MMP, 6-methylmercaptopurine.










