|
 |
- Search
| Intest Res > Epub ahead of print |
|
Abstract
Background/Aims
Methods
Results
Conclusions
ADDITIONAL INFORMATION
Funding Source
This work was supported by research grant from the Korean Society of Gastroenterology.
Data Availability Statement
The data underlying this article are available on the following website. Health Insurance Review and Assessment Service (HIRA) claim databases (https://opendata.hira.or.kr/home.do) and The Korean Statistical Information Service databases (https://kosis.kr/index/index.do).
Author Contribution
Conceptualization: Oh EH, Park SH (4th). Data curation: Oh EH, Kim YJ, Kim M. Formal analysis: Oh EH, Kim YJ, Kim M. Project administration: Park SH (6th). Resources: Oh EH, Kim YJ, Kim M. Supervision: Park SH (6th). Writing - original draft: Oh EH. Writing - review & editing: Oh EH, Kim YJ, Park SH (6th). Approval of final manuscript: all authors.
Supplementary Material
Supplementary Table 1.
Supplementary Table 2.
Supplementary Table 3.
Supplementary Table 4.
Supplementary Table 5.
Supplementary Fig. 1.
Supplementary Fig. 2.
Supplementary Fig. 3.
Supplementary Fig. 4.
Fig. 2.
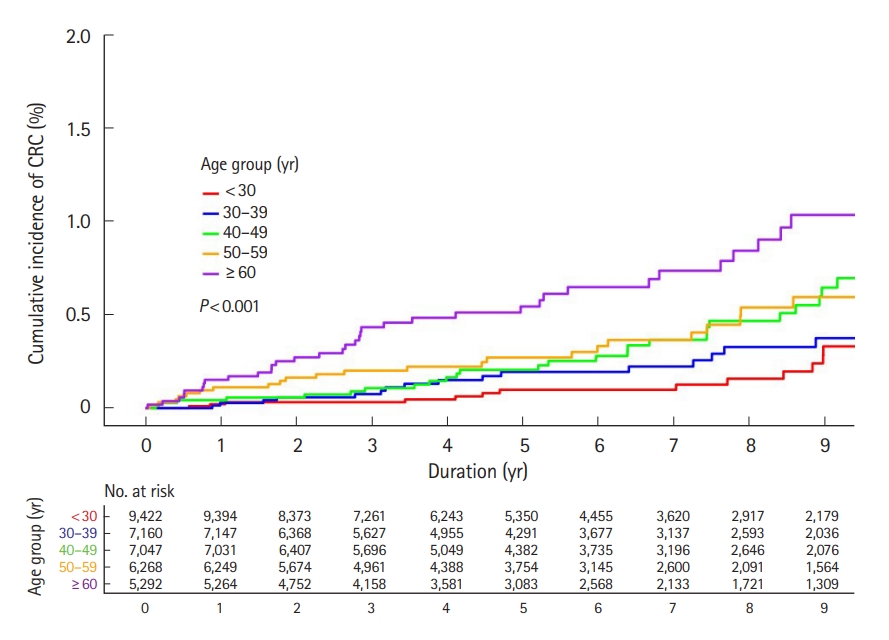
Fig. 3.

Table 1.
Table 2.
| Site |
Total (n=35,189, 100.0%) |
Male (n=21,368, 60.72%) |
Female (n=13,821, 39.28%) |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of incident cases | Sum of PY | IR (/105) | No. of expected cases | SIR (95% CI) | No. of incident cases | Sum of PY | IR (/105) | No. of expected cases | SIR (95% CI) | No. of incident cases | Sum of PY | IR (/105) | No. of expected cases | SIR (95% CI) | |
| All malignancies | 1,086 | 218,988.990 | 495.915 | 1,157.200 | 0.938 (0.883-0.996) | 610 | 131,191.440 | 464.969 | 729.843 | 0.836 (0.771-0.905) | 476 | 87,797.550 | 542.156 | 427.357 | 1.114 (1.016-1.219) |
| Lip, oral cavity and pharynx | 26 | 223,071.850 | 11.655 | 19.952 | 1.303 (0.851-1.909) | 18 | 133,346.200 | 13.499 | 16.163 | 1.114 (0.660-1.760) | 8 | 89,725.650 | 8.916 | 3.789 | 2.111 (0.909-4.160) |
| Larynx | 6 | 223,175.800 | 2.688 | 7.324 | 0.819 (0.299-1.783) | 5 | 133,416.760 | 3.748 | 7.076 | 0.707 (0.228-1.649) | 1 | 89,759.040 | 1.114 | 0.248 | 4.028 (0.053-22.410) |
| Esophagus | 11 | 226,627.260 | 4.854 | 15.494 | 0.710 (0.354-1.270) | 11 | 136,864.440 | 8.037 | 14.653 | 0.751 (0.374-1.343) | 0 | 89,762.820 | 0.000 | 0.841 | NE |
| Stomach | 97 | 222,833.300 | 43.530 | 166.402 | 0.583 (0.473-0.711) | 64 | 133,209.830 | 48.045 | 129.462 | 0.494 (0.381-0.631) | 33 | 89,623.470 | 36.821 | 36.940 | 0.893 (0.615-1.255) |
| Small intestine | 6 | 223,184.970 | 2.688 | 4.665 | 1.286 (0.470-2.800) | 4 | 133,434.300 | 2.998 | 3.292 | 1.215 (0.327-3.111) | 2 | 89,750.670 | 2.228 | 1.373 | 1.456 (0.164-5.258) |
| Colon and rectuma | 122 | 221,484.850 | 55.083 | 146.734 | 0.831 (0.690-0.993) | 81 | 132,324.200 | 61.213 | 105.527 | 0.768 (0.610-0.954) | 41 | 89,160.650 | 45.984 | 41.207 | 0.995 (0.714-1.350) |
| Liver | 83 | 222,882.870 | 37.239 | 90.811 | 0.914 (0.728-1.133) | 58 | 133,233.130 | 43.533 | 76.238 | 0.761 (0.578-0.984) | 25 | 89,649.740 | 27.886 | 14.573 | 1.715 (1.110-2.532) |
| Extrahepatic bile duct and gall bladder | 36 | 223,079.570 | 16.138 | 31.614 | 1.139 (0.797-1.577) | 23 | 133,365.230 | 17.246 | 21.115 | 1.089 (0.690-1.635) | 13 | 89,714.340 | 14.490 | 10.499 | 1.238 (0.659-2.117) |
| Pancreas | 42 | 223,068.710 | 18.828 | 33.090 | 1.269 (0.915-1.716) | 27 | 133,350.680 | 20.247 | 22.181 | 1.217 (0.802-1.771) | 15 | 89,718.030 | 16.719 | 10.909 | 1.375 (0.769-2.268) |
| Lung | 115 | 222,805.700 | 51.614 | 139.124 | 0.827 (0.682-0.992) | 85 | 133,140.310 | 63.842 | 110.163 | 0.772 (0.616-0.954) | 30 | 89,665.390 | 33.458 | 28.961 | 1.036 (0.699-1.479) |
| Thyroid | 180 | 222,345.850 | 80.955 | 149.558 | 1.204 (1.034-1.393) | 58 | 133,176.530 | 43.551 | 42.406 | 1.368 (1.038-1.768) | 122 | 89,169.320 | 136.818 | 107.152 | 1.139 (0.945-1.359) |
| Breast | 100 | 222,847.160 | 44.874 | 88.686 | 1.128 (0.917-1.371) | 2 | 133,441.170 | 1.499 | 0.558 | 3.586 (0.403-12.946) | 98 | 89,405.990 | 109.612 | 88.129 | 1.112 (0.903-1.355) |
| Cervix uteri | 23 | 223,102.580 | 10.309 | 15.248 | 1.508 (0.956-2.263) | 0 | 133,441.420 | NE | NE | NE | 23 | 89,661.160 | 25.652 | 15.248 | 1.508 (0.956-2.263) |
| Corpus uteri | 10 | 223,173.930 | 4.481 | 11.442 | 0.874 (0.418-1.607) | 0 | 133,441.420 | NE | NE | NE | 10 | 89,732.510 | 11.144 | 11.442 | 0.874 (0.418-1.607) |
| Ovary | 16 | 223,148.620 | 7.170 | 10.532 | 1.519 (0.868-2.467) | 0 | 133,441.420 | NE | NE | NE | 16 | 89,707.20 | 17.836 | 10.532 | 1.519 (0.868-2.467) |
| Prostate | 90 | 222,846.530 | 40.387 | 75.294 | 1.195 (0.961-1.469) | 90 | 133,083.710 | 67.627 | 75.294 | 1.195 (0.961-1.469) | 0 | 89,762.820 | NE | NE | NE |
| Testis | 0 | 223,204.240 | 0.000 | 1.718 | NE | 0 | 133,441.420 | 0.000 | 1.718 | NE | 0 | 89,762.820 | NE | NE | NE |
| Kidney | 38 | 223,050.910 | 17.036 | 27.953 | 1.359 (0.962-1.866) | 28 | 133,342.940 | 20.998 | 21.992 | 1.273 (0.846-1.840) | 10 | 89,707.970 | 11.147 | 5.961 | 1.678 (0.803-3.085) |
| Bladder | 30 | 223,110.820 | 13.446 | 23.778 | 1.262 (0.851-1.801) | 24 | 133,365.360 | 17.996 | 20.902 | 1.148 (0.735-1.709) | 6 | 89,745.460 | 6.686 | 2.877 | 2.086 (0.762-4.540) |
| Central nervous system | 17 | 223,129.480 | 7.619 | 9.183 | 1.851 (1.078-2.964) | 9 | 133,396.050 | 6.747 | 5.967 | 1.508 (0.688-2.863) | 8 | 89,733.430 | 8.915 | 3.216 | 2.488 (1.071-4.902) |
| Hodgkin lymphoma | 2 | 223,203.550 | 0.896 | 1.439 | 1.390 (0.156-5.018) | 1 | 133,441.230 | 0.749 | 1.045 | 0.957 (0.013-5.323) | 1 | 89,762.320 | 1.114 | 0.394 | 2.539 (0.033-14.128) |
| Non-Hodgkin lymphoma | 32 | 223,071.620 | 14.345 | 23.312 | 1.373 (0.939-1.938) | 19 | 133,374.050 | 14.246 | 16.051 | 1.184 (0.712-1.849) | 13 | 89,697.570 | 14.493 | 7.261 | 1.790 (0.952-3.062) |
| Multiple myeloma | 6 | 223,188.560 | 2.688 | 7.841 | 0.765 (0.279-1.666) | 4 | 133,429.670 | 2.998 | 5.222 | 0.766 (0.206-1.961) | 2 | 89,758.890 | 2.228 | 2.619 | 0.764 (0.086-2.757) |
| Leukemia | 20 | 223,135.640 | 8.963 | 15.495 | 1.291 (0.788-1.994) | 12 | 133,401.390 | 8.994 | 10.637 | 1.128 (0.582-1.971) | 8 | 89,734.250 | 8.915 | 4.859 | 1.647 (0.709-3.245) |
| Melanoma | 6 | 223,178.040 | 2.688 | 2.852 | 2.103 (0.768-4.579) | 3 | 133,432.470 | 2.248 | 1.746 | 1.719 (0.345-5.021) | 3 | 89,745.570 | 3.343 | 1.107 | 2.710 (0.545-7.919) |
| Other skin cancer | 31 | 223,130.570 | 13.893 | 23.834 | 1.301 (0.884-1.846) | 20 | 133,398.180 | 14.993 | 14.002 | 1.428 (0.872-2.206) | 11 | 89,732.390 | 12.259 | 9.832 | 1.119 (0.558-2.002) |